Tracing the flow of cerebrospinal fluid
Swelling is one of the most dangerous and immediate consequences of a brain injury or stroke. Doctors have long known about the dangers of swelling, which has traditionally been blamed on ruptured blood vessels. New research suggests the brain's other plumbing system, the one that circulates cerebrospinal fluid (CSF), may play an underappreciated role in both good health and response to injury.
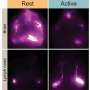
Douglas Kelley, a mechanical engineer at the University of Rochester who uses fluid dynamics to probe the inner workings of the brain, teamed up with Rochester neuroscientist Maiken Nedergaard to demonstrate the early swelling immediately after an injury or stroke results not from blood, but from an inrush of CSF. The blood flows in later through tears in the blood-brain barrier.
"There is this whole other fluid transport system beyond blood," said Kelley, who presented the work at the 73rd Annual Meeting of the American Physical Society's Division of Fluid Dynamics. "It matters for disease, and for pathology, and it matters for drug delivery."
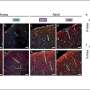
Researchers had assumed that CSF only flowed around brain tissue. Then, in 2012, Nedergaard's group published evidence pointing to the existence of CSF pathways through the brain. Their findings suggested that during sleep, CSF flows along these glymphatic pathways and rinses away cellular debris, like the amyloid-beta and tau proteins that accumulate and have been linked to Alzheimer's disease. Since then, research into the fluid dynamics of CSF has emerged as its own subfield that can provide new insights to biologists and neuroscientists.
"Having numbers on things helps you make better predictions," said Kelley. "They let us make predictions about the speed of flow, and when flow is more important, and when diffusion is more important. We can make better predictions now than anybody could three or four years ago."
Saikat Mukherjee, a postdoctoral researcher at the University of Minnesota, Twin Cities, noted that researchers still disagree about whether or not CSF enters brain tissue. If it doesn't, then the brain primarily relies on diffusion to clear toxic proteins. If CSF does seep into the brain tissue, even a little, then advection—the clearing of material by fluid flow—could help significantly with the cleanup.
The difference may be huge. "Toxic proteins get released from the brains and don't just sit there," said Mukherjee. "They aggregate into higher and higher molecular weight proteins." Mukherjee's work suggests that diffusion is not as efficient in clearing larger aggregates, while advection may clear proteins of any size. If advection does turn out to play a role, he said, then perhaps that knowledge could be harnessed to develop new neurodegenerative disease treatments that better clear protein aggregates.
Mukherjee and his colleagues are currently studying clinical data on plaque buildup in the brain to see how well it matches their simulations. They're also reviewing findings from studies investigating the clearance of toxic proteins during the sleep-wake cycle.
Ultimately, said Mukherjee, using fluid dynamics to study brain fluids points opens up two clear pathways of research. First, it can help neuroscientists better understand how the body gets rid of cellular debris—and what happens, from a physics point of view, when that system breaks down. Second, it could lead to insights on more fundamental questions about fluid dynamics and reaction-diffusion transport mechanisms in the brain.
"It lets us look at new physics that no one else has looked at yet," said Mukherjee.
More information: Brain Cerebrospinal Fluid Flow, meetings.aps.org/Meeting/DFD20/Session/C02.1
A Porous Media Model of Transport in the Brain Interstitium, meetings.aps.org/Meeting/DFD20/Session/H02.3
Provided by American Physical Society