Ultra-small hollow alloy nanoparticles for synergistic hydrogen evolution catalysis
Because hydrogen fuel has high energy density and does not pollute the environment, it has now shown the potential to replace fossil energy. The hydrogen evolution reaction (HER) is one of the most promising hydrogen production methods as a half-reaction of the electrolysis of water. Currently, traditional Pt-based compounds are used as the most active electrocatalysts for hydrogen evolution reactions. However, Pt is relatively scarce and expensive. Therefore, designing and synthesizing highly efficient, stable, and inexpensive catalysts is a frontier topic in the field of water electrolysis.
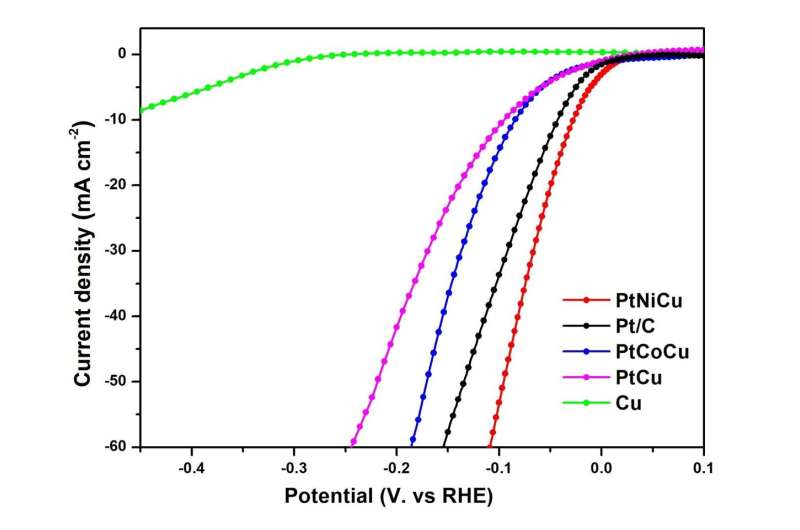
Recently, Zhenxing Li and his team from China University of Petroleum (Beijing) have made exciting progress in the preparation of HER catalysts, using a simple one-pot method to synthesize ultra-small hollow ternary alloy nanoparticles, including PtNiCu nanoparticles, PtCoCu nanoparticles and CuNiCo nanoparticles. During synthesis, the displacement reaction and oxidative etching played important roles in the formation of hollow structures. The average size of PtNiCu nanoparticles is only 5 nm and contains only 10% Pt. The unique hollow structure and large specific surface area increase the degree of surface atom exposure, provide abundant active centers, and make PtNiCu nanoparticles exhibit excellent electrocatalytic activity and stability. In alkaline electrolyte, the overpotential of hollow PtNiCu nanoparticles at 10 mA cm-2 is as low as 28 mV versus RHE with a Tafel slope of 52.1 mV dec-1, which was lower than those of commercial Pt/C. In addition, its mass activity is 5.62-fold higher than that of commercial Pt/C system. This effectively reduces the cost of platinum-based electrocatalysts and ensures that platinum atoms are used more efficiently.
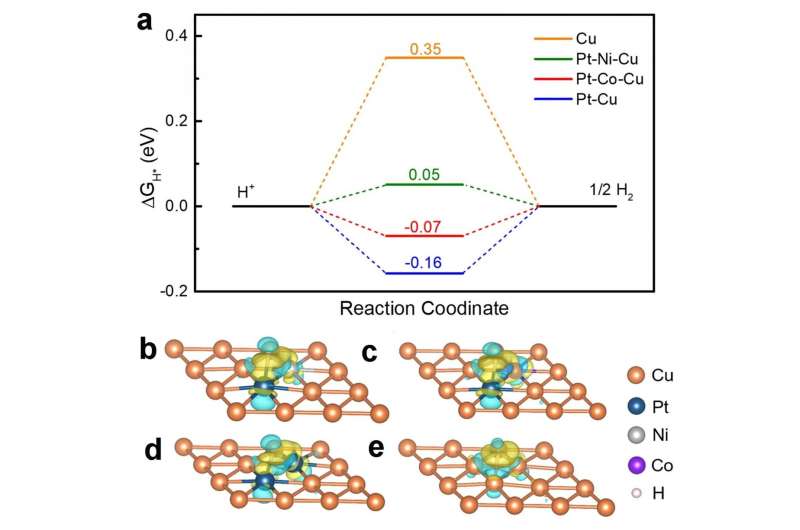
By analyzing the bonding and antibonding orbital filling, the density functional theory (DFT) calculations show that the ΔGH* of PtNiCu nanoparticles is 0.05 eV, which is close to zero. In the hydrogen evolution reaction (HER) reaction process, the bonding strength of different metals to the hydrogen intermediate (H*) was in the order of Pt > Co > Ni > Cu. Thus, the excellent HER performance of hollow PtNiCu nanoparticles can be attributed to moderately synergistic interactions between the three metals and H*. Combining theoretical calculations with experimental data, this work provides a new strategy for the design and preparation of low-cost and high-performance HER catalysts.
More information: Zhenxing Li et al, Ultra-small hollow ternary alloy nanoparticles for efficient hydrogen evolution reaction, National Science Review (2020). DOI: 10.1093/nsr/nwaa204
Provided by Science China Press