Scientists discover unusual materials properties at ultrahigh pressure

An international team of scientists from NUST MISIS (Russia), Linköping University (Sweden) and University of Bayreuth (Germany) found that, contrary to the usual physical and chemical laws, the structure of some materials does not condense at ultrahigh pressures. Actually, it forms a porous framework filled with gas molecules. This happened with samples of Os, Hf, and W put together with N in a diamond anvil at a pressure of one million atmospheres. The discovery is described in Angewandte Chemie.
"You can transform a pencil lead into diamond if you squeeze it very hard"—this fact heard by many of us in childhood sounded like complete nonsense. However, science laws make it clear that there is no miracle: both pencil lead and diamond are formed by the same chemical element, i.e. carbon, which actually forms a different crystal structure under very high pressure. But it makes sense: air pressure in the empty space between atoms decreases and the material becomes denser. Until recently, this statement could be applied to any material.
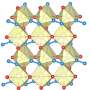
It turns out that a number of materials can become porous at ultrahigh pressure. Such a conclusion was made by a group of scientists from NUST MISIS (Russia), Linköping University (Sweden) and University of Bayreuth (Germany). The team examined three metals (hafnium [Hf], tungsten [W], and osmium [Os]) with an addition of N when placed in a diamond anvil at a pressure of 1 million atmospheres, which corresponds to a pressure at a depth of 2.5 thousand kilometers underground. Scientists believe that it was the combination of pressure and nitrogen that influenced the formation of a porous framework in the crystal lattice.
"Nitrogen itself is quite inert, and without ultrahigh pressure it would not react with these metals in any way. Materials without nitrogen would simply condense in a diamond anvil. However, a combination yielded an amazing result: some of the nitrogen atoms formed a kind of reinforcing framework in the materials, allowing the formation of pores in the crystal lattice. Consequently, additional nitrogen molecules entered the space," said Professor Igor Abrikosov, head of the theoretical research group and NUST MISIS Laboratory for the Modeling and Development of New Materials.
The experiment was initially conducted physically by the Swedish and German members of the group, and then its results were confirmed by theoretical modeling on a NUST MISIS supercomputer. Scientists emphasize that the research is fundamental, i.e. materials with such properties are not yet created for specific tasks. At the moment, the very fact that previously unthinkable modifications of materials can be obtained is important.
A whole new step will be to preserve such materials at normal atmospheric pressure. In one of the previous works, scientists managed to preserve a special modification of rhenium nitride. Currently, rapid cooling to critical low temperatures is considered as one of the ways to stabilize new materials.
More information: Maxim Bykov et al, High‐Pressure Synthesis of Metal–Inorganic Frameworks Hf 4 N 20 ⋅N 2 , WN 8 ⋅N 2 , and Os 5 N 28 ⋅3 N 2 with Polymeric Nitrogen Linkers, Angewandte Chemie International Edition (2020). DOI: 10.1002/anie.202002487
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by National University of Science and Technology MISIS