A spicy silver lining

Distinctive thermal and electrical characteristics make silver nanoparticles perfect for optics and biosensing applications. One increasingly popular application for the nanoparticles is as an antibacterial coating. Silver nanoparticle coatings are used in fabrics, footwear, computer keyboards, and orthopedic and other biomedical devices.
The process of synthesizing the silver nanoparticles involves chemical reduction of silver nitrate salt (AgNO3). A pair of reagents breaks down the AgNO3 molecules, releasing silver ions that are "reduced" through the acceptance of electrons. This favors the formation of silver nanoparticles. However, the reagents are reported to have toxic properties, so since the early 2000s, ecofriendly methods have been sought to reduce silver nitrate and other metal nanoparticles, including gold and platinum.
Researchers David Omar Oseguera-Galindo and Eden Oceguera-Contreras, both of the University of Guadalajara, Mexico, and Dario Pozas-Zepeda of the University of Colima, Mexico, recently studied the effect of habanero pepper in the synthesis of silver nanoparticles. Their research, published in the Journal of Nanophotonics, resulted in a simple, low-cost, ecofriendly method of obtaining silver nanoparticles.
Habaneros were chosen not only for their antioxidant content but also their economic importance as a traditional crop in Mexico. Previous studies showed that high antioxidant capacity promoted the formation of the nanoparticles. Other plants, such as aloe vera, coconut extract, green tea, pineapple, and garlic cloves, have been used in the synthesis of nanoparticles.
The researchers measured pH and oxidation reduction potential—a molecule's ability to gain or lose electrons—in real time to explain the kinetic performance of the nanoparticles. They also suggested an explanation about how the biomolecules may affect nanoparticle formation. Habanero pepper has evolved antioxidant machinery that decreases the effect of oxidative stress. The antioxidants are key to stabilizing molecules through reducing and scavenging electrons.
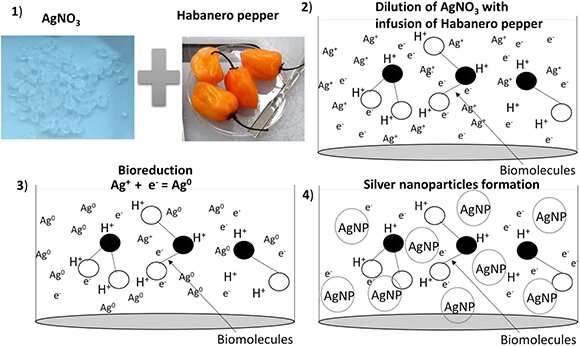
"The novelty of this research is that it offers a schematic of silver nanoparticle formation by the biosynthesis method," Oseguera-Galindo said. "In this schematic is included a possible mechanism of the biomolecules and its effect in the silver ion reduction for favoring nanoparticle formation."
Experimental results showed that samples prepared using 0.001 M and 0.005 M of AgNO3 yielded particles with an average size of 19.3 and 26.4 nm. The nanoparticles prepared with 0.005 M had an absorption peak shifted toward a larger wavelength, which was expected because these nanoparticles also had a bigger average size.
In a second experiment, the researchers used varying concentrations of habanero (10, 50, and 100 mL), monitoring the pH and oxidation reduction potential. The 100-mL sample had the most intense absorption peak and the highest pH decrease, indicating the most nanoparticle production. This sample had more biomolecules, consistent with more proton removal, which would increase the number of electrons available for the reduction of silver ions.
Thanks to this green method of synthesizing silver nanoparticles, habanero peppers may soon be used to synthesize the antimicrobial silver nanoparticles in your next pair of running shorts or that next squirt of antibacterial hand sanitizer, without toxic effects.
More information: David Omar Oseguera-Galindo et al, Silver nanoparticles synthesis using biomolecules of habanero pepper (Capsicum chinense Jacq.) as a reducing agent, Journal of Nanophotonics (2020). DOI: 10.1117/1.JNP.14.036012
Provided by SPIE