Researchers find therapeutic targets to fight SARS-CoV-2
Researchers from HSE University have developed new approaches for regulating the expression of ACE2 and TMPRSS2 enzymes, which play a crucial role in cell infection with SARS-CoV-2. The scholars discovered that small non-coding microRNA (miRNA) molecules are capable of performing a targeted decrease in ACE2 and TMPRSS2. The results of the study have been published in PLOS ONE.
Angiotensin-convertingenzyme 2 (ACE2) and transmembrane serine protease 2 (TMPRSS2) enzymes act as entrance gates into the cell for the novel coronavirus. After successful penetration, the virus uses the cell's recourses to replicate and exit the cell to infect new cells. Research teams all over the world are experimenting with medicinal impact on ACE2 and TMPRSS2 aimed at blocking opportunities for SARS-CoV-2 virus to enter human cells.
In addition, the ACE2 enzyme also plays a major role in the development of the acute respiratory distress syndrome, the main cause of death in patients with COVID-19. Apart from the respiratory organs, ACE2 and TMPRSS2 are also present in other parts of the body, such as the digestive system, kidneys and liver. This explains the variety of symptoms in patients, including gastrointestinal issues.
Stepan Nersisyan and Alexander Tonevitsky from the HSE Faculty of Biology and Biotechnology, together with their colleagues from Hertsen Moscow Oncology Research Center (Maxim Shkurnikov), Shemyakin-Ovchinnikov Institute of Bioorganic Chemistry RAS (Evgeny Knyazev) and German Cancer Research Center, Heidelberg (Andrey Turchinovich), looked at potential approaches for influencing the abovementioned enzymes via miRNA molecules.
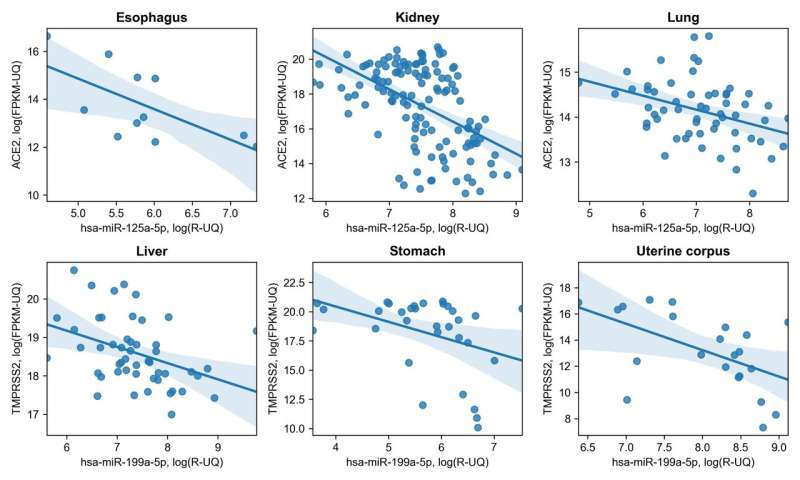
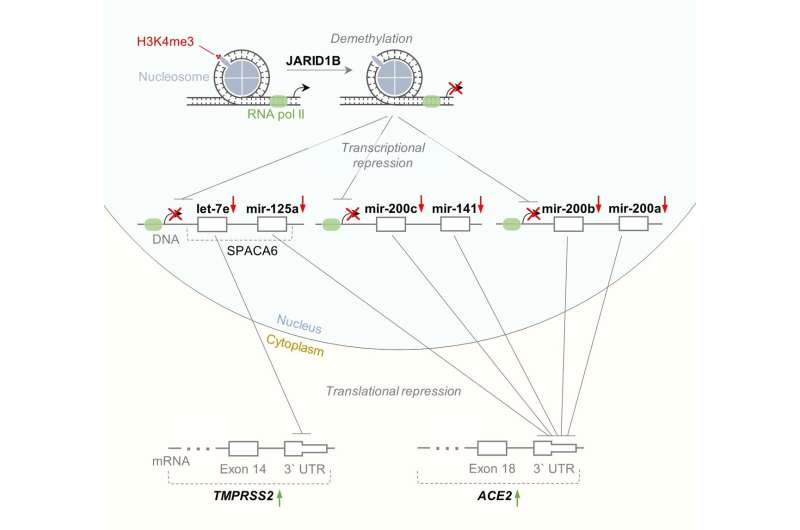
They carried out bioinformatic analysis of publicly available datasets on RNA sequencing in tissues of ten key human organs. The main task was to find miRNAs, the expression of which would demonstrate a significant negative correlation with ACE2 and TMPRSS2 gene expression. This means that the more miRNA, the less ACE2/TMPRSS2, and vice versa. As a result, the researchers detected a certain amount of such interactions that are specific for several organs at the same time. In particular, they discovered that lysine-specific demethylase 5B (JARID1B) can indirectly affect ACE2/TMPRSS2 expression by repressing transcription of hsa-let-7e/hsa-mir-125a and hsa-mir-141/hsa-miR-200 miRNA families which are targeting these genes.
More information: PLOS ONE (2020). journals.plos.org/plosone/arti … journal.pone.0235987
Journal information: PLoS ONE
Provided by National Research University Higher School of Economics