Researchers map mechanisms in the largest CRISPR system
The largest and most complex CRISPR system has been visualized by researchers from the University of Copenhagen in a new study. The system may have potential applications in biomedicine and biotechnology, the researchers believe.
CRISPR technology can be used to edit genes and revolutionized the scientific world when it was first introduced. CRISPR-Cas9 is likely the most known CRISPR system and popularly known as the "gene scissors."
That is just one out of the many CRISPR systems that exist. Now, researchers from the University of Copenhagen (UCPH) have mapped and analyzed the atomic structure of one of the most complex CRISPR systems identified to date.
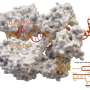
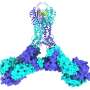
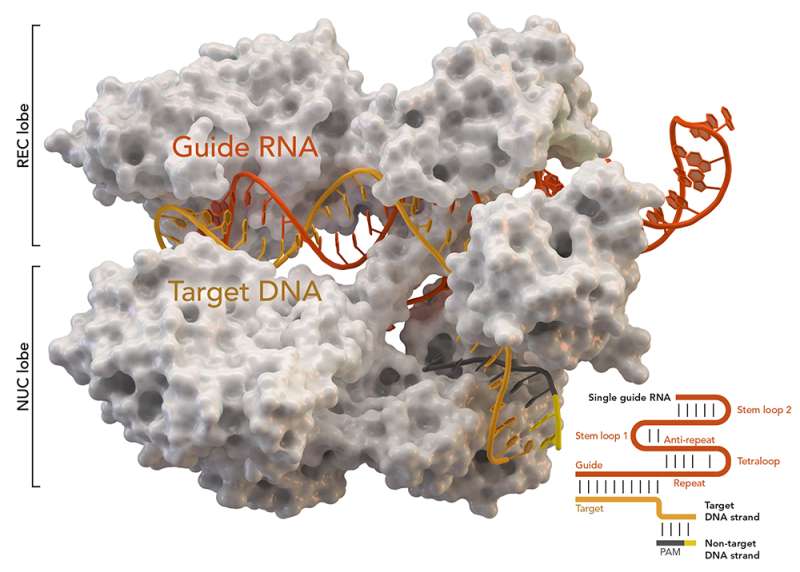
"We have solved the largest and most complicated CRISPR-Cas complex seen so far. We now understand how this system works on a molecular level," says co-author Guillermo Montoya, who is Professor at the Novo Nordisk Foundation Center for Protein Research (NNF CPR), UCPH. The researchers have studied a complex called Cmr-β, which belongs to the subgroup of so-called type III-B CRISPR-Cas complexes. The new results have been published in the scientific journal Molecular Cell.
Fights against phages
CRISPR is a system found in bacteria, among other organisms, and it is involved in bacteria's immune system. Here it plays a main role in the constant fight against invading phages, a virus that attacks bacteria.
In the new study, the researchers have studied Cmr's role in the immune system and delved into the mechanisms behind its immune response against phages and how it is regulated.
"Our findings, in collaboration with the She group at the Faculty of Sciences, highlight the diverse defense strategies of type III complexes. We have also identified a unique subunit called Cmr7, which seems to control the complex activity, and we further believe that it may defend against prospective viral anti-CRISPR proteins," says co-author Nicholas Heelund Sofos, postdoc at NNF CPR.
Potential applications
The Cmr system mapped by the researchers in the new study can, among other things, remove single-stranded RNA and DNA, though it will be very difficult to use like CRISPR-Cas9 for gene editing as it is too big and complex. But in the future, it may still be key to understanding the immune response of bacteria, and it could have some use in the fight against antibiotic resistance.
"This complex plays an important role in the fight between bacteria and phages. Antibiotic resistance comes from this type of fight. Therefore, our results may constitute important knowledge for fighting antibiotic resistance. The complex may also have therapeutic potential. In the future, we may be able to use this for diagnostics or a health problem we may not even have seen yet. Now, our goal is to look for an application for this system," says Guillermo Montoya.
The researchers used the advanced technology cryo electron microscopy—also called CryoEM—to outline the system. All research and data collection was conducted at the University of Copenhagen.
More information: Molecular Cell (2020). DOI: 10.1016/j.molcel.2020.07.008
Journal information: Molecular Cell
Provided by University of Copenhagen