Imaging of sequential potential distribution changes in electrodes during charge/discharge
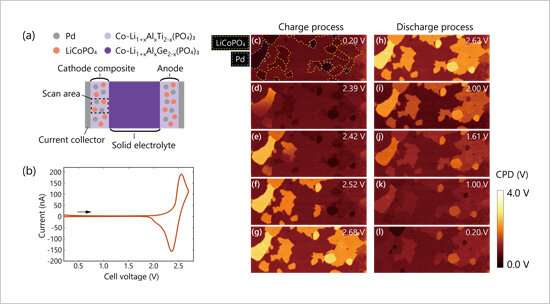
NIMS has succeeded for the first time in visualizing sequential changes in electrical potential distribution across a composite electrode during charge/discharge reactions in all-solid-state lithium ion batteries. This distribution was previously measureable only before and after the occurrence of charge/discharge reactions. Microscopic understanding of charge/discharge reaction mechanisms in electrodes may facilitate the design of new devices capable of improving the performance of all-solid-state lithium ion batteries.
The all-solid-state lithium ion battery is a promising next-generation battery candidate because of its excellent safety and cyclic properties. Its performance needs to be further improved before it can be put into practical use. The causes of performance degradation need to be identified through close analysis of electrochemical reactions occurring in electrodes during charge and discharge. In 2016 this research team developed a technique enabling in-situ observation of electrical potential changes in electrodes at high spatial resolution. However, a great deal of interest existed in the development of new techniques that would enable more detailed analyses: sequential (dynamic) measurement of changes in electrical potential distribution across an electrode during charge/discharge.
This research team recently developed a new technique capable of visualizing sequential (dynamic) changes in electrical potential distribution in batteries by combining an electrochemical measurement system with previously developed techniques (in-situ Kelvin probe force microscopy and a technique used to prepare cross-sectional battery samples). The team then used this new combined technique to observe charge/discharge reactions in action within composite cathodes in all-solid-state lithium ion batteries (experimental samples supplied by Taiyo Yuden Co., Ltd.). As a result, the team found that charge reactions extend unevenly across the composite cathode from the current collector side to the anode side, whereas discharge reactions spread evenly across the cathode. These results indicate the formation of an inadequate electronic conductive path network in the composite cathode during a charge process.
The technique developed in this research is applicable to a variety of battery evaluations, including in-depth analysis of the causes of battery performance degradation, which have been difficult to ascertain using conventional electrochemical measurements. This technique may help battery developers optimize battery structures and design higher performance batteries.
More information: Hideki Masuda et al. Dynamically visualizing battery reactions by operando Kelvin probe force microscopy, Communications Chemistry (2019). DOI: 10.1038/s42004-019-0245-x
Provided by National Institute for Materials Science