Researchers find a new HIT defense bacteria use against antibiotics
Scientists at the Severinov Laboratory in Skoltech and their colleagues from Russia and the U.S. have uncovered a new mechanism of bacterial self-defense against microcin C, a potent antibiotic weapon in the microscopic world that can sometimes turn on its master.
Microcin C is a peptide-nucleotide antibiotic produced by some strains of Escherichia coli. It is essentially a Trojan horse: Its peptide component helps it get into a cell, where the cell's own internal machinery turns it into what's called "processed McC." This compound completely blocks protein biosynthesis by interfering with its crucial component, aspartyl-tRNA synthetase.
Unfortunately for the microcin C producer, some of the "Greeks" in this metaphor inevitably escape from the "horse" too early, while it is still inside the producing cell, which leads to self-intoxication. That is why the producing cell has to get creative in developing defenses against its own weapon; one of these defenses is an enzyme that acetylates processed McC, rendering it useless.
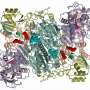
Skoltech Ph.D. student Eldar Yagmurov and his colleagues have found another way cells can protect themselves—histidine-triad (HIT) superfamily hydrolases—that is, enzymes that break a larger molecule into smaller ones using water.
"HIT hydrolases have long been suspected to be involved in the mechanisms of self-defense against microcin C. One particular enzyme in this family is known to break the bond between phosphorus and nitrogen that connects the two parts in a complex very similar to McC—so we figured there might be some other member of the superfamily that can work against microcin C," says Yagmurov.
The researchers used bioinformatics to predict a cluster in the genome of Hyalangium minutum, a Gram-negative bacterium, that encodes the production of its McC-like compounds and a particular HIT superfamily phosphoramidase that they suspected might provide self-immunity to these antibiotics. Experiments showed that this was indeed true: the enzyme apparently destroys the bond between the "transport" and "warhead" parts of processed McC, deactivating the latter.
"By studying the naturally existing means of antibiotic resistance, especially for a promising antimicrobial agent such as McC, we can try to be one step ahead of the bacteria and modify the antibiotic in a way that would help it evade these natural defenses," Yagmurov adds.
According to the paper, other bacteria may have their own analogues of the H. minutum HIT enzyme, each protecting against a specific McC-like compound that they use to survive in the tough bacterial world. This also implies a plethora of yet-unidentified McC-like compounds, some of which may have the potential to become practically used antibiotics of the future.
More information: Eldar Yagmurov et al, Histidine-Triad Hydrolases Provide Resistance to Peptide-Nucleotide Antibiotics, mBio (2020). DOI: 10.1128/mBio.00497-20
Journal information: mBio
Provided by Skolkovo Institute of Science and Technology