How blood cells deform, recover when traveling through tiny channels
Laboratory blood tests are often done by forcing samples through small channels. When the channels are very small, as in microfluidic devices, red blood cells (RBCs) are deformed and then relax back to their original shape after exiting the channel. The way the deformation and relaxation occur depends on both the flow characteristics and mechanical properties of the cell's outer membrane.
In this week's issue of the journal Biomicrofluidics, a method to characterize the shape recovery of healthy human RBCs flowing through a microfluidic constricted channel is reported. This investigation revealed a coupling between the cell's mechanical properties and the hydrodynamic properties of the flow. In addition, the method could distinguish between healthy RBCs and those infected by the malaria parasite. This suggests a possible new technique for diagnosing disease.
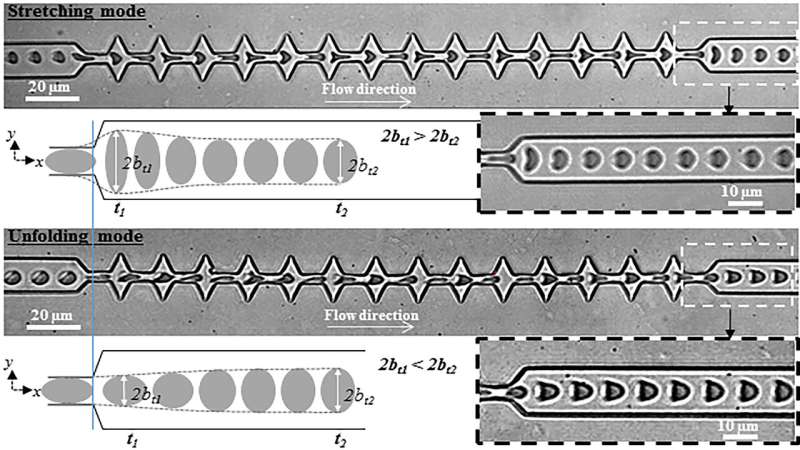
The microfluidic device consisted of a narrow channel interspersed by a succession of sawtooth-shaped wider areas. A solution of RBCs is pumped through the system by applying pressure from one end. As the cells travel through the channel, they are observed with a microscope. The images are captured with a high-speed camera and sent to a computer for analysis.
When an RBC enters a narrow channel, it takes on a parachutelike shape. When it exits into a wide region, it elongates in the direction of the flow until it meets the next widening and is again stretched by the flow.
At the final exit, two different shape recovery behaviors were observed, depending on the flow speed and viscosity of the medium. At high flow speed and viscosity, the cells get stretched upon their last exit from the channel and then recover their original shapes. At lower speed and viscosity, however, the parachutelike shape is recovered directly upon exiting.
The investigators found that the hydrodynamic conditions at which the transition between these two different recovery behaviors occurs depend on the elastic properties of the RBC.
Co-author Magalie Faivre said, "Although the time necessary for the cells to recover their shape after exiting the channel was shown to depend on the hydrodynamic conditions, we have demonstrated that, at a given stress, this recovery time can be used to differentiate healthy from Plasmodium falciparum-infected RBCs." Plasmodium falciparum is one of the parasites that cause malaria.
The investigators are seeking to expand their study to find a way to detect "signatures" for other types of diseases.
"We are currently evaluating if our approach is able to discriminate the alteration of different structural components of the RBC membrane," said Faivre. "To do so, we are studying RBCs from patients with malaria, sickle cell anemia and hereditary spherocytosis."
More information: "Dual shape recovery of red blood cells flowing out of a microfluidic constriction," Biomicrofluidics (2020). aip.scitation.org/doi/10.1063/5.0005198
Journal information: Biomicrofluidics
Provided by American Institute of Physics