Success in synthesizing black phosphorus with a safe and high-yielding method toward artificial photosynthesis

A joint research group at Osaka City University and Sakai Chemical Industry Co., Ltd. have succeeded to synthesize black phosphorus with one-pot solvothermal reaction with red phosphorus in ethylenediamine used as a solvent.
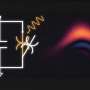
Recently, black phosphorus has been a candidate for photocatalyst working under visible light. However, the synthesis of black phosphorus is usually done at high temperature and pressure, causing difficulties to make large quantities of it. The research group discovered a condition for synthesizing black phosphorus with high yield from red phosphorus and demonstrated its high photocatalytic activity for hydrogen evolution. Their report was published in the March 12 issue of Journal of Material Chemistry A.
Photocatalytic water splitting has attracted attention as a solution to control global warming. Although various metal oxides have been used as photocatalysts, most of them do not work with visible light due to their wide band gap. Recently, black phosphorus has been studied as a candidate photocatalyst working under visible light irradiation. Black phosphorus is a layered semiconductor having a narrow band gap which can be altered by changing its layer thickness. The synthesis of black phosphorus is usually done at high temperatures and pressure that makes it difficult to synthesize higher quantities. It was necessary to develop a method for synthesizing black phosphorus by wet chemical approach suitable for making large quantities for it. Recently, some researchers reported that solvothermal reaction can convert white phosphorus to black phosphorus. As white phosphorus is very dangerous and toxify, it was important to develop synthesis method using red phosphorus safer than white phosphorus as a starting material.
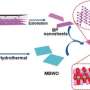
In this study, the team applied one-pot solvothermal reaction with ethylenediamine as a solvent and red phosphorus, safe to handle and widely used in industry, as a starting material to synthesize black phosphorus.
The team revealed a mechanism of black phosphorus formation by various spectroscopies.
By optimizing the concentrations and particle sizes of red phosphorus, the reaction temperature, and time, the team succeeded in synthesizing black phosphorus. Content of black phosphorus in the sample with conventional methods are around 10% but the team increased them to 90%.
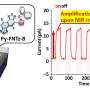
Furthermore, synthesized black phosphorus with cocatalyst showed high photocatalytic activities for hydrogen evolution from methanol aqueous solution under visible light irradiation.
Phosphorene, a single layer of black phosphorus, is attracting attention as a novel semiconductor material because of having a band gap in contrast with graphene.
This high-yielding method of black phosphorus is useful not only in the field of photocatalyst but also electronic materials.
More information: Akiyo Ozawa et al. Black phosphorus synthesized by solvothermal reaction from red phosphorus and its catalytic activity for water splitting, Journal of Materials Chemistry A (2020). DOI: 10.1039/C9TA13441G
Journal information: Journal of Materials Chemistry A
Provided by Osaka City University