Viruses reprogram cells into different virocells
If it looks like a duck and quacks like a duck, so the adage goes, it must be a duck. But if the duck gets infected by a virus so that it no longer looks or quacks like one, is it still a duck? For a team led by researchers from The Ohio State University and the University of Michigan studying how virus infections cause significant metabolic changes in marine microbes, the answer is no. They refer to the infected microbial cells as virocells, a change in name first described in 2011 which reflects the metabolic changes they've undergone.
The number of microbes in, on, and around the planet is an astronomical figure, and yet viruses outnumber them. In the ocean, viruses outnumber surface microbes 10 to 1, and 20% to 40% of the microbes are infected at any given time. Microbial viruses can thus have significant impacts on the global nutrient cycles regulated by their hosts. For example, microbes that capture and store carbon in the ocean could fix less carbon when infected. Little is known about virus-infected microbial cells that are transformed into virocells, and how the outcomes of these infections can affect the interactions within their ecosystems.
Microbes drive the energy and nutrient cycles that fuel the planet, and viruses control them through infection, but little is known about the impacts of these infections on ecosystem functions. As part of efforts to characterize global microbial diversity with an aim to improve predictive ecosystems models, University of Michigan's Melissa Duhaime and The Ohio State University's Matt Sullivan used an experimental approach with a marine phage-host model to study virus-host-nutrient interactions. In their study, which appeared in the ISME Journal, they focused on how virus-infected microbes are transformed into virocells that are metabolically and functionally different from uninfected cells. Duhaime and Sullivan are co-corresponding authors, while the study's lead author is Cristina Howard-Varona, a postdoctoral researcher in Sullivan's lab.
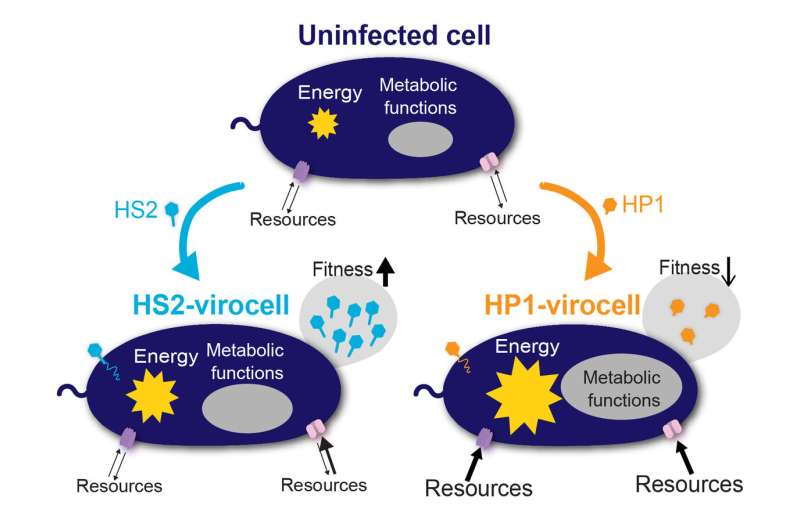
The team infected the marine Pseudoalteromonas bacterium with two unrelated viruses: siphovirus PSA-HS2 and podovirus PSA-HP1. Each virus demonstrated a different approach to infecting and metabolically reprogramming the bacterial host, which was transformed into two very different virocells—HS2-virocell and HP1-virocell, respectively—and correspondingly different infection outcomes. reprogrammed the bacteria's central carbon and energy metabolism, to obtain more nutrients from the environment.
Demonstrating that the costs of infecting microbes and their impacts on the ecosystems can vary is a step toward developing more efficient predictive ecosystems models. Next, the researchers are working towards better understanding how nutrient availability, or lack thereof, impacts viral infection and the metabolism of the resulting virocells, to better represent how infections occur in natural settings.
More information: Cristina Howard-Varona et al. Phage-specific metabolic reprogramming of virocells, The ISME Journal (2020). DOI: 10.1038/s41396-019-0580-z
Journal information: ISME Journal
Provided by DOE/Joint Genome Institute