Scientists create listeriosis-immune mice by turning off gene in myeloid cells

An international research team that includes specialists from ITMO University has conducted a series of experiments with the goal of studying the immune system and identifying the genes and proteins involved in the response to certain harmful bacteria. The scientists found that "turning off" a gene responsible for the production of the protein Beclin 1, or the gene that produces the FIP200 protein, resulted in the test animals becoming nearly completely immune to the infectious disease listeriosis. The results of this research have been published in Nature Microbiology.
The way the immune system functions is an extremely complex process and the mechanics of it have still not been fully understood by scientists. Which proteins are responsible for the immune response? Can an organism be made genetically impervious to all, or at least some malignant bacteria? Biologists and geneticists all over the world seek answers to these questions.
In order to understand how the immune system works, scientists conduct experiments and study mice that possess certain non-functioning genes as a result of prolonged targeted cross-breeding. When this approach, known to scientists as "gene knockout," results in changes to the way an animal's immune system operates, it means that the gene has a direct effect on the immune response.
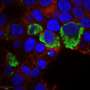
An international research team has conducted a study of mice that have had two genes "knocked out," namely the ones responsible for the production of two proteins: Beclin 1 and FIP200. Notably, the knockout was made not to the entire organism, but to the myeloid cells (a sub-type of immune cells) of the mice. These proteins are involved in the process of autophagy, which is the degradation of molecules and organelles by the cells. It was found that this knockout made the animals almost completely impervious to the Listeria monocytogenes bacteria—the cause of the dangerous disease listeriosis. The disease affects the nervous system and, if left untreated, may be fatal.
"Listeria monocytogenes is a bacterium that divides within the cell," explains Konstantin Zaitsev, a co-author of the study, Ph.D. student, and staff member of ITMO University's Computer Technologies Lab. "We've found that disabling the genes responsible for Beclin 1 or FIP200 allows the mice to fight off the infection and recover nicely."
Such a result has to do with the fact that the immune system of a mouse with knocked-out genes is activated permanently, as opposed to regular mice whose immune systems are only activated by external factors.
Macrophages, a type of white blood cells, are therefore constantly excited, which allows them to instantly take action when Listeria monocytogenes is introduced to the organism.
At first glance, gene editing doesn't seem to have any fatal effect on the test animals' lives: they grow, develop, feed, and procreate as normal. But that doesn't mean that turning off their genes leaves the mice unscathed. As the experiments showed, other infections, such as influenza, still affect the test mice at the same rate as regular mice. It should also be noted that, while the test animals developed nearly in the same manner as their regular counterparts, they were vulnerable to their own immune system: the scientists registered signs of chronic inflammation in some of the mice's organs.
"A permanently active immune system is a double-edged sword," explains Zaitsev. "There is a lot of background inflammation going on. It's no accident that the immune system is at rest most of the time. We don't want it reacting to the many processes that take place within the organism."
Nevertheless, the researchers' study provides a great deal of analytical material in regards to the effects of proteins Beclin 1 and FIP200 on the immune response to pathogenic microorganisms.
More information: Ya-Ting Wang et al, Select autophagy genes maintain quiescence of tissue-resident macrophages and increase susceptibility to Listeria monocytogenes, Nature Microbiology (2020). DOI: 10.1038/s41564-019-0633-0
Journal information: Nature Microbiology
Provided by ITMO University