The greenbacks in mobile phone mines
No-one who is economically active can afford to go without at least a mobile phone, and at the pace that electronic equipment is re-invented, it's only a matter of time before your 'latest' iPhone 11 ends up on a dump site.
As fast as we produce electronic equipment, we produce electronic waste. In fact, according to a joint report by the Platform for Accelerating the Circular Economy (PACE) and several United Nations agencies under the umbrella of the E-waste Coalition, the world produces 50 million tonnes of electronic and electrical waste (e-waste) per year—this is more than the weight of all commercial airliners ever made.
And this electronic and electrical waste has a value of over US$62,5 billion.
Mine mobile, not Earth
As the world's population creeps closer to 10 billion people on Earth by 2050, the amount of e-waste we generate increases concurrently. Although we're well on track to producing 120 million tonnes of e-waste per year by 2050, there is literally a silver lining in the amount of e-waste we produce: As much as 7 percent of the world's gold may be contained in e-waste.
The idea, says Professor Dean Brady, Head of the School of Chemistry at Wits, is to create a circular economy, where everything that is used can be recycled, reused, or repurposed into new products. "We have a lot of rare earth metals in phones that we can re-use," says Brady.
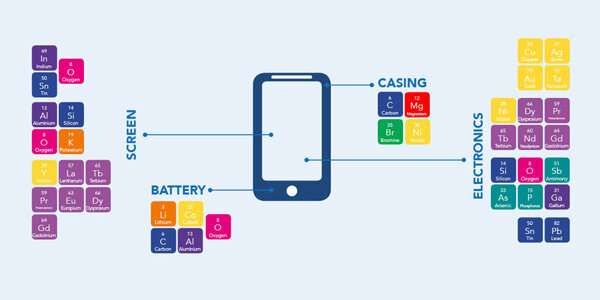
According to the PACE joint report, there is 100 times more gold in a tonne of e-waste than there is in a tonne of gold ore. Furthermore, our cell phones, laptops, solar panels, and other electronic devices are crammed with rare earth materials such as platinum, cobalt, magnesium and copper.
Economy of circularity
"Almost everything from your phone can be recycled," says Brady. "Our cell phones are going to become the next mines. If we can take all the rare earth metals out of our phones rather than of out of the ground, we can reuse them and create a circular economy."
Currently only 20 percent of all e-waste is recycled, with the rest either ending up on a landfill or being recycled informally. In a circular economy, almost everything could be re-used or repurposed, leaving very little to go to waste.
Brady and his colleagues are working towards closing the recycling gap, by creating a circular economy from the chemical processes required to mine cell phones.
"While chemistry has changed the world during the last 150 years and has been responsible for major advances in almost every industry, we as chemists are also highly conscious of the impact we have on the environment," says Brady.
Inspired by Wits Professor Roger Sheldon, who is widely known as the "Father of Green Chemistry", Wits chemists are exploring green chemistry practices through researching renewable chemicals, recoverable catalysts for sustainable chemical processes, using CO2 and green chemistry through using enzyme reactions for anything from biofuels to pharmaceuticals and food ingredients.
"There is a huge wealth disparity across the world, and the great demand for more and cheaper products implies that we would need to manufacture about four times what we are manufacturing currently. However, we already have the sustainable [carbon] footprint of 1.7 Earths [we are currently using the resources of 1.7 Earths to sustain ourselves], so we are going to need to produce about three to four times as much as we currently do with half the environmental footprint we now have," says Brady. "This is a tall order."
The School of Chemistry is working on new technologies such as green chemistry through renewable chemicals; finding recoverable catalysts for chemical reactions through using CO2 as a catalyst and using biocatalysts to produce anything from biofuels to pharmaceuticals and food ingredients.
The alchemy of green chemistry
Professor Charles de Koning, lead on the project to find renewable chemicals, says: "Crude oil is the basis of our chemicals industry, but this is not sustainable. Not only is the supply of crude oil finite, but our use of fossil fuels is releasing carbon deposited over millennia into the atmosphere as carbon dioxide, which will result in climate change."
The School of Chemistry is developing and using new products derived from biomass to replace fossil fuel-based chemicals, which will result in neutral net CO2 production.
"New technologies in chemical catalysis, bio-catalysis and biotransformation, flow chemistry, and bio-refineries will contribute to a more sustainable circular economy, with reduced waste, as defined by the Environmental (E)-Factor developed by Professor Roger Sheldon," says Brady.
Ultimately, this green chemistry can galvanise a circular economy that can recycle cell phones to greenbacks. Sustainably.
Provided by Wits University




















