November 12, 2019 feature
Mechanisms of soft tissue and protein preservation in Tyrannosaurus rex

The existing notion that soft tissue architectures and native proteins can be preserved across geological time is controversial since methods of such preservation remain to be investigated and well-defined. In a new study, Elizabeth M. Boatman and colleagues at the departments of Engineering, Paleontology, Biological Science, Materials and Engineering and the Advanced Light Source in the U.S., tested crosslinking mechanisms for preserved tissue architecture. They used two non-enzymatic, structural protein mechanisms, Fenton chemistry and glycation to demonstrate their possible contributions to preserve blood vessel structures recovered from the cortical bone of Tyrannosaurus rex (T. rex; USNM 555 000, formerly MOR 555). They demonstrated the endogeneity (randomness) of the fossil vessel tissues and the presence of type I collagen in the outermost vessel layers using imaging, diffraction, spectroscopy and immunohistochemistry.
They derived data from synchrotron Fourier transform infrared (SR-FTIR) studies on the T. rex vessels to analyze their crosslinking character and compared them with control chicken samples treated similarly with the two techniques. The researchers provided X-ray microprobe analyses of the chemical state of the fossil tissues to support vessel preservation of T. rex, as observed using the methods of investigation. Boatman et al. propose that the observed tissue stabilizing crosslinks will play an important role to preserve additional microvascular tissues in skeletal elements from the Mesozoic era. The work is now published on Scientific Reports.
Paleontologists have recovered hollow, pliable and transparent vessel-like structures from skeletal elements of fossil vertebrates including non-avian dinosaurs and applied many techniques to identify their endogenous proteins such as collagen and elastin. Researchers had used mass-spectroscopy sequencing to identify isolated vessels recovered from non-avian dinosaurs to support the presence of vertebrate-specific vascular proteins in the past. For example, they documented the hallmark 67-nanometer-banding pattern typical for type I collagen after liberating the protein via demineralization, followed by additional studies to verify the presence of type I collagen in vascular canals of a sauropod dinosaur rib from approximately 190 million years ago using FTIR and Raman analysis. While research teams had developed a variety of methods to explain unexpected preservation, experimental testing of proposed mechanisms remain to be conducted routinely and broadly.
In the present work, Boatman et al. identified and tested the possible contribution of a set of experiments to preserve the vessel-like architecture of the compact bone of a Tyrannosaurus rex fossil. They expect the work to lay a possible foundation for additional studies on preserving soft tissues recovered from the Mesozoic or more recent fossils. The walls of vertebrate blood vessel contain three distinct layers including the tunica intima (innermost), tunica media and tunica externa (outermost layer). Due to their unique molecular compositions, scientists can differentiate the constituents morphologically and chemically. For example, elastin is a helical protein specific to vertebrates that offers resistance to pressure changes in the vascular walls. Collagen is also vertebrate-specific and constitutes a predominant fraction of blood vessels to serve as their structural foundation. Since elastin and collagen contain hallmark features identifiable at the molecular structure and composition, Boatman et al. proposed to study the two proteins within the remnant dinosaur vessels.
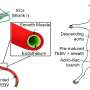
![LEFT: SR-FTIR analysis. Amide I sub-band localization of untreated and treated chicken type I collagen in SR-FTIR spectra. Sub-bands (β-sheet, ~1633 cm−1; triple-helix, ~1658–1660 cm−1; intermolecular, ~1683–1690 cm−1) are indicated in the figures. Red traces denote second derivatives of experimental curves. Although the intermolecular sub-band typically presents at lower wavenumber, the identified value was the nearest local minimum in each of the second derivative traces and consistently appears across all samples; therefore, in this sample, the intermolecular sub-band was indexed at 1697–1699 cm−1. RIGHT: SEM images of USNM 555000 cortical bone. (a) Fracture surface showing clear features of osteons (o) predominantly in longitudinal section, osteocyte lacunae (ol; in dashed white circles), and fine texture consistent with mineralized collagen fibers in bone. Back-scattered (BSE) image. (b), Polished (1200 grit) transverse section (BSE image) showing clear features of osteons and osteocyte lacunae. Mineral infilled osteons (white arrows) yield highly altered vessel structures, which were readily eliminated from SAXS, FTIR, and TEM analysis by careful preparation (sedimentation, washing, selection under microscope). Cracks are due to humidity/pressure changes and are an artefact of preparation. (c), Polished (1200 grit) transverse section (secondary electron [SE] image) showing clear features of osteons and osteocyte lacunae. (d), Highly magnified SE image of an osteon, showing fibrous texture at edges (white arrow), which was commonly observed in non-mineral infilled osteons in this specimen. This thin, fibrous coating inside the osteon structure is proposed to be the hollow, pliable vessel structures. Credit: Scientific Reports, doi: 10.1038/s41598-019-51680-1 Mechanisms of soft tissue and protein preservation in Tyrannosaurus rex](https://scx1.b-cdn.net/csz/news/800a/2019/1-mechanismsof.jpg)
The research team hypothesized the contribution of early diagenetic (physical and chemical) processes to the survival of T. rex microvasculature from deep-time. To test this, Boatman et al. first conducted SR-FTIR analysis to understand crosslink character in their control sample of chicken type I collagen protein. They induced crosslinks in the protein using Fenton reagent or ion-catalyzed glycation techniques followed by the use of transmission SR-FTIR to test each tissue. They observed the intramolecular crosslinks formed in the chicken tissues to be immature due to their lack of exposure to pathways necessary to form intermolecular crosslinks or advanced glycation endproducts (AGEs).
To test the T. rex vessel architecture for endogenous proteins, the scientists liberated three types of vessels from a demineralized T. rex cortical bone. They then used visible light microscopy (VLM) to characterize them as:
- Extensive, brown-hued pliable networks
- Fragmented opaque structures
- Fragmented semi-translucent structures
They coupled energy-dispersive X-ray spectroscopy (EDS) with scanning electron microscopy (SEM) as well as micro-focused X-ray fluorescence (µXRF) spectroscopy to confirm the differences observed in the tissue samples of varying composition. The team focused on the pliable vessel networks due to their similarly to existing bone tissue, which presumably maintained minimal alteration.
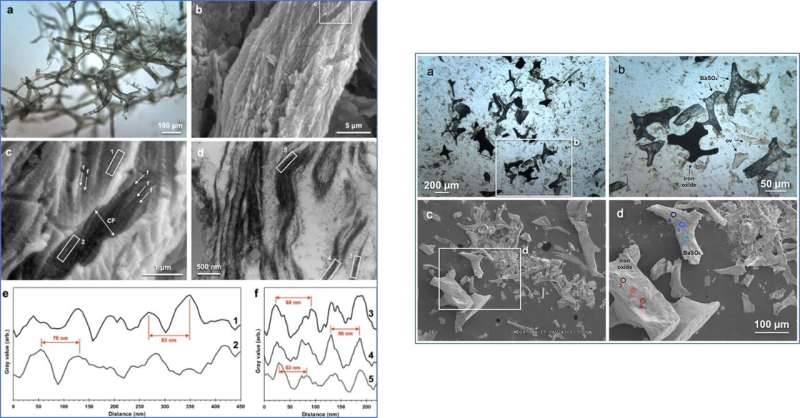
When Boatman et al. studied the pliable T. rex vessels using SEM, they observed fibrous structures across their outermost surface. The combined features were consistent with those observed in extant vessels liberated from cortical bone and with fibrillar collagen. The team analyzed the SR-FTIR spectrum of T. rex vessels to detect the dominant bands observed in both treated extant and ancient tissues. Notably, the amide I band for the dinosaur tissue was located at a predominant α-helix structure consistent with mature (crosslinked) fibrillar collagen. The research team then conducted immunohistochemistry (IHC) studies to identify protein-specific epitopes of the structural proteins elastin and type I collagen.
The scientists raised antibodies against all components of the extant vasculature to observe positive binding in the dinosaur vessel walls. Using a fluorescent filter, they captured the localization and distribution of antibody-antigen complexes (green fluorescence). The response of the dinosaur vessels to actin antibodies appeared as a thin and evenly distributed layer. Antibodies raised against the muscle protein tropomyosin appeared with greater intensity on the vessel walls. The dinosaur vessels also indicated the presence of type I collagen antibodies, although elastin antibodies showed greater intensity. The two proteins were good targets for fossil studies due to high evolutionary conservation in certain regions. They did not observe reactivity of dinosaur vessels to antibodies against bacterial peptidoglycan (indicating no microbial contamination).
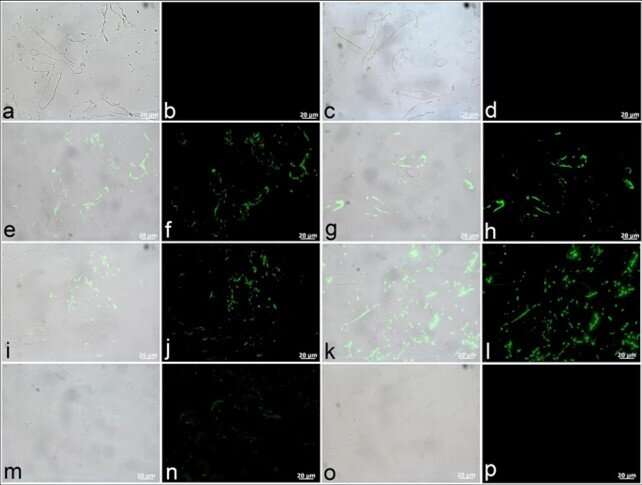
Boatman et al. tested T. rex vessel structures to understand if post-mortem structural protein crosslinking enhanced their resistance to degradation or diagenetic alterations. For this, they focused on fibrillar collagen using SR-FTIR transmission spectra to suggest post-mortem crosslinking during the process of tissue architecture preservation. These spectral features were previously recorded but not discussed with early Jurassic sauropodomorphs and cretaceous bones. The scientists then treated bulk T. rex tissue with sodium borohydride (NaBH4) to reduce carbonyl groups within immature crosslinks and increase the non-peptide carbonyl absorption intensity. The carbohydrate absorption bands in the T. rex tissue were consistent with AGEs (advanced glycation endproducts). After treatment, the data suggested that T. rex tissues possessed both intramolecular and intermolecular crosslink types.
When the scientists mapped the elements in the tissue using µXRF, they revealed iron (Fe) as the only metal concentrated within the dinosaur vessel tissues while recording barium (Ba) within the semi-translucent vessel casts. Using extended micro X-ray absorption near-edge structure microscopy, they observed Fe3+ embedded in the vessel walls. The researchers showed the presence of finely crystalline goethite (α-FeO(OH)); a mineral previously detected in vascular tissues recovered from two diverse dinosaur specimens.
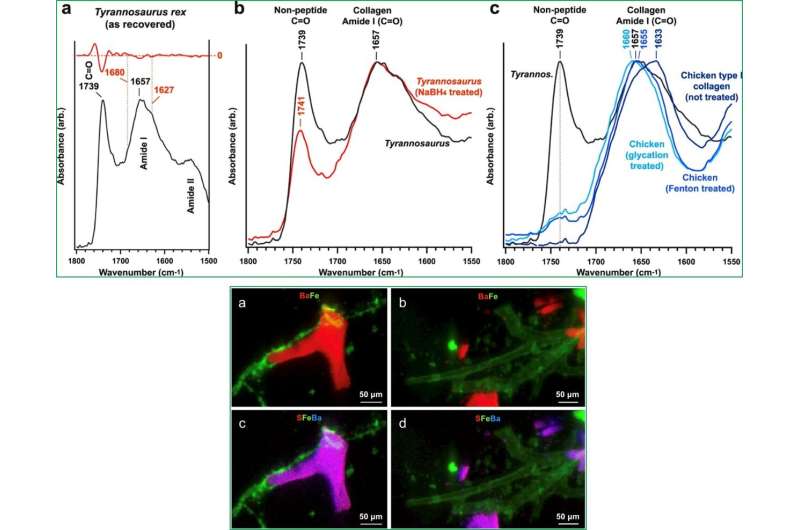
In this way, Elizabeth M. Boatman and colleagues demonstrated the presence of vertebrate-species endogenous proteins within soft tissue dinosaur structures. This included the presence of type I collagen consistent with the vasculature in extant vertebrates. The data supported a two-step mechanism that stabilized biomolecules and vessel architecture after the death of the organism, to promote their preservation within skeletal elements. The team hypothesized that iron-mediated Fenton and glycation pathways may have contributed to enhanced T. rex tissue longevity of elastin and fibrillar collagen within and around blood vessels. Both processes could be catalyzed by transition metal species such as iron to define the central role of Fe observed in structural protein crosslinking. The formation of iron oxyhydroxide precipitates in the work fully supported this idea.
The data represent the first comprehensive chemical and molecular characterization of vascular tissues recovered from T. rex specimen USNM 555000. The results shed light on the possible processes of fossilization at the molecular level. The researchers envision the demonstrated techniques will contribute to the development of comprehensive mechanisms to consistently retain vascular tissue survival from deep time.
More information: Elizabeth M. Boatman et al. Mechanisms of soft tissue and protein preservation in Tyrannosaurus rex, Scientific Reports (2019). DOI: 10.1038/s41598-019-51680-1
Mary H. Schweitzer et al. A role for iron and oxygen chemistry in preserving soft tissues, cells and molecules from deep time, Proceedings of the Royal Society B: Biological Sciences (2013). DOI: 10.1098/rspb.2013.2741
Sergio Bertazzo et al. Fibres and cellular structures preserved in 75-million–year-old dinosaur specimens, Nature Communications (2015). DOI: 10.1038/ncomms8352
Journal information: Scientific Reports , Proceedings of the Royal Society B , Nature Communications
© 2019 Science X Network