Study investigates a critical transition in water that remains liquid far below the freezing point

Water can remain liquid at temperatures far below 0 degrees Celsius. This supercooled phase is a current focus for scientific research. A theoretical model developed at São Paulo State University (UNESP) in Brazil shows that in supercooled water, there is a critical point at which properties such as thermal expansion and compressibility exhibit anomalous behavior.
Led by Mariano de Souza, a professor in the Physics Department of UNESP's Institute of Geosciences and Exact Sciences at Rio Claro, the study was supported by FAPESP. An article by Souza and collaborators describing the study has been published in Scientific Reports.
"Our study shows that this second critical point is analogous to the liquid-gas transition in water at about 374 degrees Celsius and at a pressure of some 22 megapascals," Souza told.
Liquid and gas phases coexist in water at approximately 374 degrees Celsius. The genesis of this exotic behavior can be observed, for example, in a pressure cooker. At this point, water's thermodynamic properties begin to display anomalous behavior. For this reason, the point is considered "critical."
In the case of supercooled water, two phases also coexist, but both are liquid. One is more dense and the other less dense. If the system continues to be cooled appropriately below 0 degrees Celsius, there comes a point on the phase diagram where the stability of the two phases breaks down, and the water starts to crystallize. This is the second critical point, determined theoretically by the recent study.
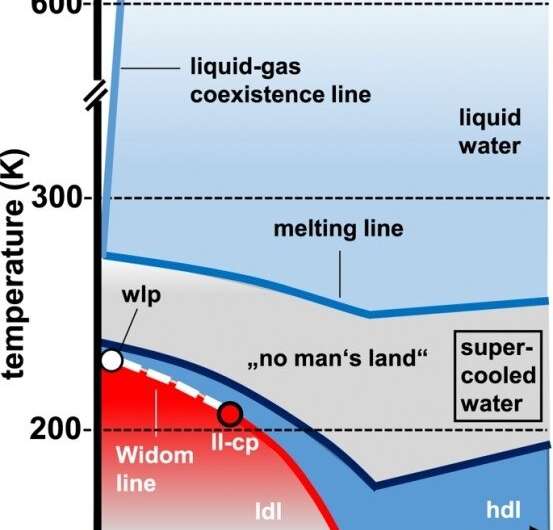
"The study shows that this second critical point occurs in the range of 180 kelvins [approximately -93 degrees Celsius]. Above this point, liquid water can exist. It's called supercooled water," Souza said.
"The most interesting part is that the theoretical model we developed for water can be applied to all systems in which two energy scales coexist. For example, it applies to an iron-based superconductor system in which there is also a nematic phase [with molecules oriented in parallel lines but not arranged in well-defined planes]. This theoretical model originated in several experiments with thermal expansion at low temperatures performed in our research laboratory."
This universal model was obtained by means of a theoretical refinement of the Grüneisen parameter, named for German physicist Eduard Grüneisen (1877-1949). Simply put, this parameter describes the effects of variations in temperature and pressure on a crystal lattice.
"Our analysis of the Grüneisen and pseudo-Grüneisen parameters can be applied to an investigation of critical behavior in any system with two energy scales. It suffices to make appropriate adjustments to the critical parameters in accordance with the system of interest," Souza said.
More information: Gabriel O. Gomes et al, Enhanced Grüneisen Parameter in Supercooled Water, Scientific Reports (2019). DOI: 10.1038/s41598-019-48353-4
Journal information: Scientific Reports
Provided by FAPESP





















