Molecular nanocarbons with mechanical bonds
Carbon materials with nano-scale periodicity such as graphene and carbon nanotubes, called "nanocarbons," are expected to become light, highly functional next-generation materials. There have been demands for precise synthesis methods targeting only at the nanocarbon structure with the desired property because their electronic and mechanical properties are greatly different depending on the structure. Under such circumstances, "molecular nanocarbon science," in which organic synthesis is used to precisely synthesize molecules with partial structures of nanocarbons, has recently received attention and thus a lot of research projects are under way all over the world.
Until now, a lot of molecules having partial structures of fullerenes, graphene and carbon nanotubes (molecular nanocarbons) have been synthesized. They have, however, relatively simpler structures from the viewpoint of topology. On the other hand, a large number of nanocarbons with a complex topology such as donut-shaped ones (torus) or coil-shaped ones have been predicted from the standpoint of theoretical chemistry, and therefore researchers have been interested in the unknown properties of such nanocarbons. As the first step of precise synthesis of such nanocarbons, a team led by Yasutomo Segawa, a group leader of the JST-ERATO project, and Kenichiro Itami, the director of the JST-ERATO project and the center director of ITbM has proposed "topological molecular nanocarbons," which are molecular nanocarbons with a complex topology.
In this research, they succeeded in synthesizing molecular nanocarbons with knots and catenanes, which are basic types of topology. Molecules called catenanes and knots have been synthesized since 1960s and 1980s, respectively. In recent years, such molecules have been expected to be applied to molecular machines (nano-scale machines) and are well known as the reason for award of the Nobel Prize in chemistry in 2016. However, to generate structures of knots or catenanes, it was necessary to introduce nitrogen atoms or oxygen atoms and induct the structure to a topological one using such atoms as a steppingstone. Therefore, a new synthesis method had to be developed for synthesizing molecular nanocarbons with knots or catenanes.
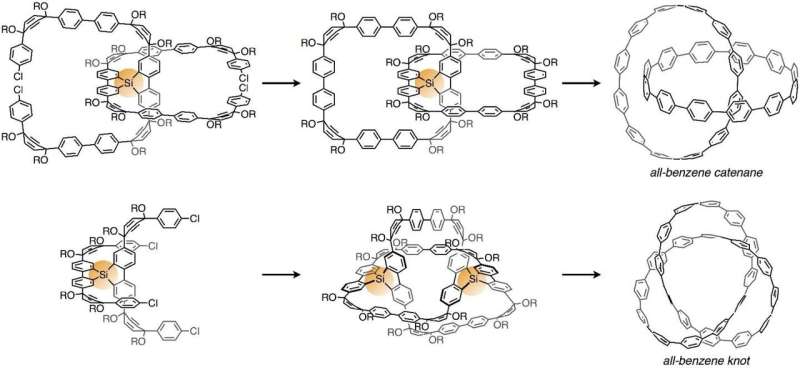
A molecular nanocarbon "cycloparaphenylene," a partial structure of a carbon nanotube, is a ring-shaped molecule with a diameter of approximately 1 nanometer that is composed only of benzenes. They thought that they would be able to introduce knots or catenanes by using silicon atoms as tentative fastenings in the middle of synthesizing cycloparaphenylenes. As those silicon atoms can be removed later, knots or catenanes composed only of carbon skeletons can be obtained in the end.
First, prepare C-shaped molecules and connect the center of two C-shaped molecules with a silicon atom. Second, connect the ends of each of those C-shaped molecules through a reaction with nickel is used to generate two rings. Third, use fluorine (tetrabutylammonium fluoride) to remove the silicon atom. Finally, a reaction with sodium is used to convert into a molecule an "all-benzene catenane" in which two cycloparaphenylenes are geometrically bound. With this synthesis method, they have succeeded in synthesizing catenanes from a pair of rings composed of 12 benzenes. Using a similar method, they have synthesized 2 milligrams of catenanes in which two rings of different sizes are bound, one consisting of 12 benzenes and the other 9 benzenes.
Further applying this synthesis method, they have synthesized topological molecular nanocarbons with knots, "all-benzene knots," which can be called "unattainable molecules" due to the greater difficulty. As other prior researches have revealed that a topology of molecular knots can be generated by arranging two tentative fastenings at appropriate positions, they designed a precursor with two silicon atoms as the tentative fastenings. They have succeeded for the first time in synthesizing all-benzene knots targeting "carbon knots" by synthesizing molecules in which U-shaped molecules were bound with silicon atoms and then processed with this unit in the same way as all-benzene catenanes (homo-coupling reaction, fluoridization, and sodium reduction reaction). X-ray crystallography was used to confirm that the molecule has a knot. In addition, they have proved the existence of carbon nanotori (donut-shaped nanocarbons) incorporating the all-benzene knot our research group has synthesized as a partial structure from the standpoint of computational science and have shown that the synthesis of all-benzene knots is an important step toward the synthesis of topological nanocarbons.
Next, they have proved that those newly synthesized molecules have specific properties derived from the knots or catenanes. It was observed that after catenanes composed of two rings that are different in size are excited by light, the excitation energy is transferred from the larger ring to the smaller ring very rapidly. The catenane structure is the only way to verify the effect of the interaction between the rings with the symmetry of each ring perfectly maintained. The experiments this time have proved that the rings electronically interact via the catenane structure.
On the other hand, when they dissolved all-benzene knots in an organic solvent to conduct NMR measurements of hydrogen nuclei, only one type of signal was observed even at a low temperature of minus 95 degrees. This indicates that signals are leveled due to very rapid motion. A simulation on a supercomputer strongly suggested that such fast-speed levelling is caused by donut-shaped vortex-like motions. They found those properties, which are very difficult to predict in advance, for the first time through synthesis and isolation.
Knots, which can be divided into left-handed ones and right-handed ones, have a property called chirality (e.g. chirality between the left hand and the right hand, which are not equivalent as they are, but become equivalent when reflected by a mirror). They have succeeded in separating the all-benzene knots we synthesized this time into the left-handed knots and right-handed knots and to prove that all-benzene knots show circular dichroism (a phenomenon of different absorption intensities between light rotating in a right-hand sense (right circular polarization) and light rotating in a left-hand sense (left circular polarization), which are observed in molecules with chirality) derived from the chirality of the knots.
Further development
The product of this research will be a big step toward the synthesis of nanocarbons with complex geometric structures. The capability of constructing complex geometric structures such as knots and catenanes from carbon skeletons will lead to the design and synthesis of unprecedented complex nanocarbons. In addition, the product can be called a monument to be included in textbooks of organic chemistry as an example of synthesizing very beautiful molecules with an innovative method. It is an epoch-making product that can be a starting point of the development of new chemistry because it has the potential to completely change the design of molecular machines based on geometric bond structures.
More information: Yasutomo Segawa et al, Topological molecular nanocarbons: All-benzene catenane and trefoil knot, Science (2019). DOI: 10.1126/science.aav5021
Journal information: Science
Provided by Japan Science and Technology Agency