Chemists create self-assembling material with suite of new properties
Chemists from have created a new material that self-assembles into 2-D networks in a predictable and reproducible manner. The material has a suite of new properties, which means it may have numerous applications—although it will take time and significant exploration to determine how it may best be used.
Importantly, the research provides a very rare example of "bottom-up" material creation; it is extremely challenging to exert control over a self-assembling material such that chemists can reliably predict and reproduce its structure dependent on the environment it is in—but the Trinity team has done just that.
Additionally, scientists have long been interested in developing anion-templated self-assemblies as they possess great potential to remove hazardous and polluting molecules from the environment. However, working with anions (ions with a negative charge) rather than cations (ions with a positive charge) is molecularly challenging for a number of reasons.
Senior author of the research, Thorfinnur Gunnlaugsson, Professor of Chemistry at Trinity, said:
"Anions are widespread in our world, with many of them carrying out specific roles in nature—for both living and inanimate matter. But, because these processes are often mediated by specificity, when any changes occur in these interactions the results can be of harm to life and the environment. For this reason, we have always been interested in gaining a deep understanding of how these molecules bond with the ultimate goal of imitating the way proteins and enzymes interact with anions in nature.
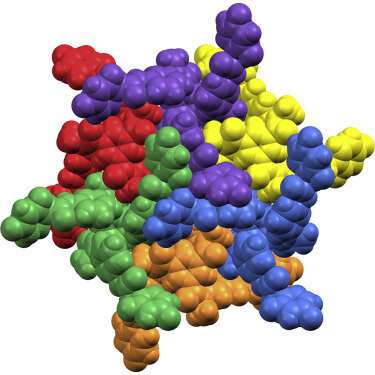
Trying to create a material that does exactly what you think it will—and what you need it to—in different environments is incredibly challenging because environments are rarely always stable. It is something of a dark art, but after a huge amount of work we have successfully created something that forms a controlled, hierarchical 2-D network, and we are able to predict exactly how it will look in different environments."
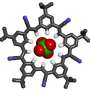
Building on their earlier work, the team 're-shaped' a ligand (a substance that forms a complex with a molecule to serve a biological purpose) by tinkering with its molecular structure so that instead of capturing sulphate ions and holding them in cage-like structures, it instead uses them as glue to make their highly ordered 2-D material.
Their ground-breaking work was supported by Science Foundation Ireland and involved a collaboration with researchers at the MacDiarmid Institute for Advanced Materials and Nanotechnology at the University of Canterbury. It is described in internationally acclaimed journal Chem.
The team is now excited to explore the properties of the new material so as to consider potential applications in the wider world. It is possible that it could make an impact in health via targeted drug delivery (it is biologically compatible); in printing or in a setting that uses gels; or even in the world of electronics, where new materials are being touted as the holding the key to longer-lasting batteries and improved performance of high-value goods.
Professor Mick Morris, Director of the SFI AMBER research center hosted in Trinity, added:
"The potential of this work cannot be undervalued. It represents the work of many years and people in Professor Gunnlaugsson's laboratory to evolve chemical methods to synthesize complex materials by design—thus allowing them to be applied in many fields. This work is an important part of our program in AMBER, allowing the center to tackle challenges we may once have found impossible."
More information: Anna B. Aletti et al. Sulfate-Templated 2D Anion-Layered Supramolecular Self-Assemblies, Chem (2019). DOI: 10.1016/j.chempr.2019.06.023
Journal information: Chem
Provided by Trinity College Dublin