Nanoparticles in lithium-sulphur batteries detected with neutron experiment
Lithium-sulphur batteries are regarded as one of the most promising candidates for the next generation of energy storage devices. They have a theoretical gravimetric energy density that is five times higher than that of the best lithium-ion batteries currently available. And they even work at sub-zero temperatures of down to -50 °C. In addition, sulphur is inexpensive and environmentally friendly.
However, their capacity so far has fallen sharply with every charge-discharge cycle, so that such batteries are not yet long-lasting. The loss of capacity is caused by complicated reaction processes at the electrodes inside the battery cell. It is therefore particularly important to understand exactly how the charge (sulphur) and discharge (lithium sulphide) products precipitate and dissolve. While sulphur precipitates macroscopically and therefore lends itself to examination by imaging techniques or X-ray diffraction during cycling, lithium sulphide is difficult to detect due to its sub-10-nm particle size.
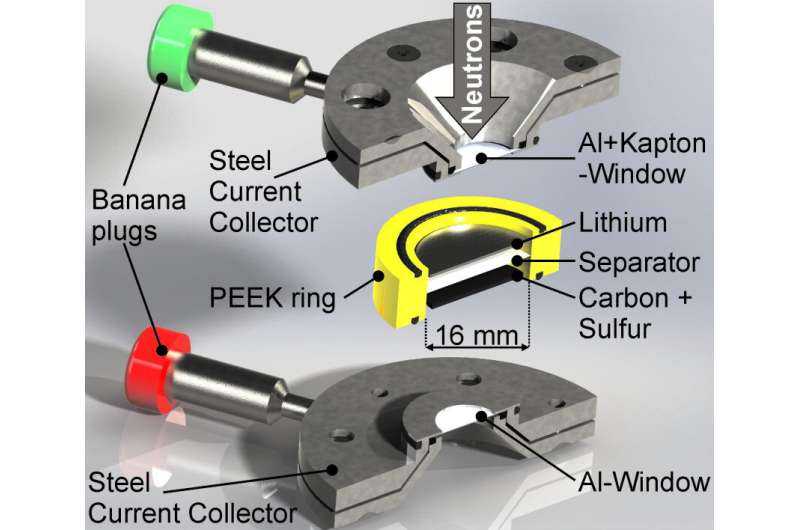
Insight into this has now been provided for the first time by investigations with the BER II neutron source at the HZB. Dr. Sebastian Risse used a measuring cell he developed to illuminate lithium-sulphur batteries with neutrons during charging and discharging cycles (operando) and simultaneously performed additional measurements with impedance spectroscopy.
This enabled him and his team to analyse the dissolution and precipitation of lithium sulphide with extreme precision during ten discharge/charging cycles. Since neutrons interact strongly with deuterium (heavy hydrogen), the researchers used a deuterated electrolyte in the battery cell to make both the solid products (sulphur and lithium sulphide) visible.
Their conclusion: "We observed that the lithium sulphide and sulphur precipitation does not take place inside the microporous carbon electrodes, but instead on the outer surface of the carbon fibres," says Risse. These results provide a valuable guide for the development of better battery electrodes.
More information: Sebastian Risse et al, Operando Analysis of a Lithium/Sulfur Battery by Small-Angle Neutron Scattering, ACS Nano (2019). DOI: 10.1021/acsnano.9b03453
Journal information: ACS Nano
Provided by Helmholtz Association of German Research Centres