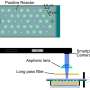
September 3, 2019 feature
Turning a handheld smartphone into a fluorescence microscope

Researchers in the U.S. and China have developed a method to transform a smartphone into a fluorescence microscope. The handheld smartphone-fluorescence microscope (HSFM) device allows complex biomedical analyses both rapidly and inexpensively. Conventional fluorescence microscopes play an important role to detect diverse cells and proteins, but they are bulky and inconvenient for point-of-care diagnoses. Now writing in Light: Science & Applications, Bo Dai and an interdisciplinary research team detailed the use of liquid polymers to create miniature two-droplet lenses dyed with colored solvents. The lenses were compatible across several different smartphone cameras. The low-cost, experimental setup allowed them to observe and count cells, monitor the expression of fluorescently tagged genes and distinguish between normal tissues and tumors. The easily accessible and affordable smartphone technology can contribute to frugal science and will lead to better administration of onsite and economically viable personalized medicine.
Fluorescence microscopy is ubiquitous in multiple disciplines, including cell and molecular biology, the healthcare industry, environmental monitoring and food sanitation. In biomedicine and clinical applications, fluorescent imaging can detect and track cells, proteins and other molecules of interest with high sensitivity and precision. Conventional fluorescence microscopes are typically engineered with bulky components, which render them extremely challenging for point-of-care diagnosis in resource limited regions. As a result, portable microscopes are an important development on an ideal smartphone platform for mobility and accessibility to a range of users.
Researchers had previously used smartphone-based microscopes to image human blood cells, waterborne parasites and human cytomegalovirus. For these research efforts they included key elements such as light-emitting diodes (LEDs) for illumination, external lenses for optical imaging and magnification as well as fluorescence emission filtering to route light. Polymer lenses are easy to develop and provide high-resolving power to build a 'do-it-yourself' microscope for resource-limited applications. However, due to the diverse models of smartphones currently available, researchers aim to develop an attachment for smartphone-based microscopy whose design is independent from a specific phone model.
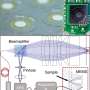
To address this challenge in the present work, Dai et al. developed a low-cost handheld smartphone fluorescence microscope (HFSM) in a portable size. The HRSM used a single compact and multifunctional color lens to convert any smartphone model into a fluorescence microscope without modifying the attachment design between phones. The experimental design reduced the HRFM device complexity and allowed its adoption across a variety of smartphones. The product is functionally consistent across multiple smartphone platforms, easy to operate, low cost, and can be mass produced. The research team used the device to demonstrate bright field and fluorescent imaging in several bioanalytical applications within cells and tissues.
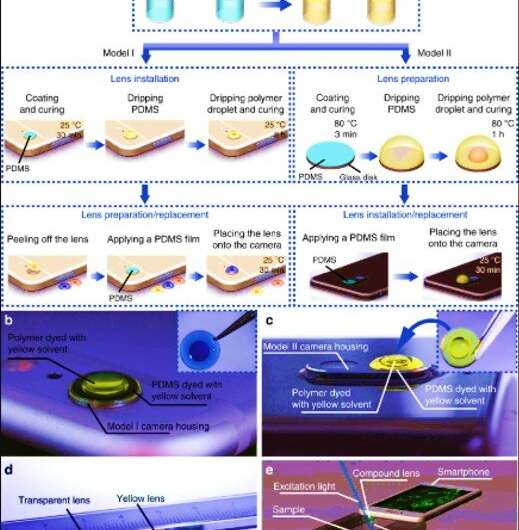
For the HFSM module, Dai et al. included a color compound lens for both imaging and light filtering. They developed the miniature lens using two high-refractive-index droplets, one inside another dyed with colored solvents to transmit the desired emission light to the imaging sensor. The researchers developed two models in the study to either (1) protrude from the back of the phone (model I) or (2) remain in profile with the phone (model II). For both versions, they included a lens design with colored polydimethylsiloxane (PDMS) pre-polymer and methyl phenyl polymer (vinyl-terminated dimethyl diphenyl siloxanes). To determine how the polymer droplet spread during the fabrication process, the researchers calculated the radius of the droplet and capillary length.
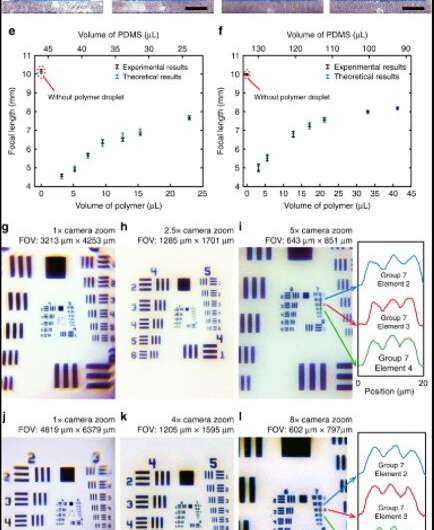
They first tested and detected the PDMS droplet to form a spherical cap under the influence of the interfacial tension force and took several factors into consideration to determine the internal and external curvature of the PDMS cap. Thereafter, when they equipped the smartphone with lenses made of 3.2 µL polymer droplets, the camera could resolve a 2.76 µm line. Since the liquid-state polymer droplet remained sealed completely within the stable and cured PDMS cap, the research team avoided problems associated with external mechanical vibrations and thermal disturbances or chemical deteriorations during its use. They adhered the lens to the camera as part of the smartphone to carry around conveniently, and could peel off the lens from the camera to replace it with a different customized lens for imaging.
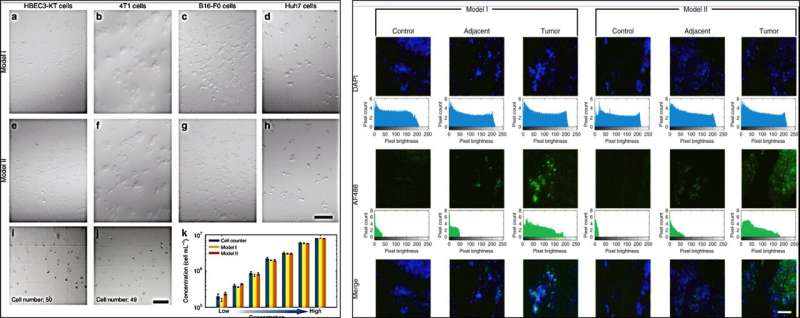
The research team further developed and employed a custom illumination tool in the microscopic imaging process to observe and count cells under white light illumination. Using the setup, they viewed cuboidal and spindle-shaped cell aggregates in small clusters. During cell counting experiments, Dai et al. clearly distinguished the individual cells and calculated the cell concentration, which agreed excellently with the results obtained from a commercial cell counter to validate the HSFM device. Thereafter, the scientists incubated human liver tissues with fluorescently tagged antibodies to detect normal or defective features using the HSFM equipped with a green lens. Using the smartphone microscope, Dai et al. accurately identified images of normal tissues, para-tumor tissues and cancer tissues. For instance, a higher expression of bright green fluorescence confirmed the presence of abnormal, diseased tissue.
The research team then used the HSFM with a green lens to monitor transfection and the expression of enhanced green fluorescent protein (EGFP; reporter gene to study physiological processes) within a plasmid. For this, they transfected the GFP-tagged human NLRP3 gene into a 293T human embryonic kidney cell line and excited the transfected cells with a 480 nm blue light for bright green fluorescence emission. The excitation light filtered through the green lens for fluorescence emission, which Dai et al. captured as green spots using the smartphone. The results agreed well for both lens models (model I and II) relative to values measured using a conventional microscope.
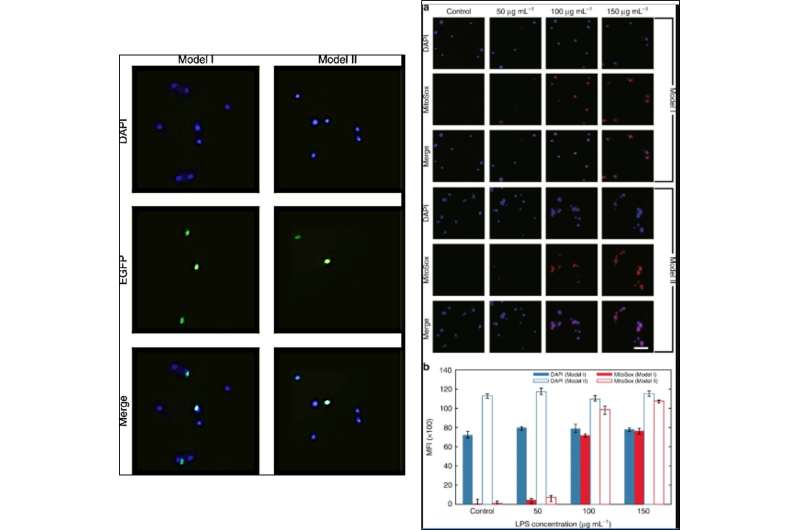
Dai et al. subsequently used the setup to quantify superoxide production; a physiological marker of cardiovascular and neurodegenerative disease. For this, they stained a HBEC3-KT human bronchial epithelial cell line with MitoSox Red, a fluorogenic probe that can highly selectively detect superoxide, which they produced by interacting HBEC3-KT cells with lipopolysaccharides (LPS) in this work. The team observed a consistent increase in the mean fluorescence intensity of MitoSox Red to support the enhanced production of superoxide after LPS triggering.
In this way, Bo Dai and co-workers provided a compact, affordable platform for fluorescence microscopy using a lens-based smartphone. The setup captured images at cellular resolution and a field of view (FOV) on a tissue-wide scale. The capabilities relied on the pixel and image sensor size within the smartphone; a technology which continues to evolve. The research team were inspired by preceding research work on a smartphone lens named the DOTlens developed elsewhere. The work presented here can serve as next-generation multifunctional lens modules for field-portable smartphone microscopes. Dai et al. believe the observed applications are merely the tip of the iceberg with more potential for future applications with the HSFM device. They expect to develop the color compound lenses for additional fluorescent channels to significantly enhance the capabilities of the cost-effective microscope. The scientists envision the mass manufacture of low-cost, simple HFSM devices for mobile and customized healthcare applications at the point of care.
More information: Bo Dai et al. Colour compound lenses for a portable fluorescence microscope, Light: Science & Applications (2019). DOI: 10.1038/s41377-019-0187-1
Yu-Lung Sung et al. Modeling the surface of fast-cured polymer droplet lenses for precision fabrication, Applied Optics (2018). DOI: 10.1364/AO.57.010342
Rainer Pepperkok et al. High-throughput fluorescence microscopy for systems biology, Nature Reviews Molecular Cell Biology (2006). DOI: 10.1038/nrm1979
Journal information: Light: Science & Applications
© 2019 Science X Network