September 9, 2019 feature
Genome engineering with CRISPR/HDR to diversify the functions of hybridoma-produced antibodies
Bioengineers and life scientists incorporate hybridoma technology to produce large numbers of identical antibodies, and develop new antibody therapeutics and diagnostics. Recent preclinical and clinical studies on the technology highlight the importance of antibody isotypes for therapeutic efficacy. In a new study, a research team in Netherlands have developed a versatile Clustered Regularly Interspaced Short Palindrome Repeats (CRISPR) and homology directed repair (HDR) platform to rapidly engineer immunoglobin domains and form recombinant hybridomas that secrete designer antibodies of a preferred format, species or isotype. In the study, Johan M. S. van der Schoot and colleagues at the interdisciplinary departments of immunology, proteomics, immunohematology, translational immunology and medical oncology, used the platform to form recombinant hybridomas, chimeras and mutants. The stable antibody products retained their antigen specificity. The research team believes the versatile platform will facilitate mass-scale antibody engineering for the scientific community to empower preclinical antibody research. The work is now published on Science Advances.
Monoclonal antibodies (mAb) have revolutionized the medical field with applications to treat diseases that were once deemed incurable. Hybridoma technology is widely used since 1975 for mAb discovery, screening and production, as immortal cell lines that can produce large quantities of mAbs for new antibody-based therapies. Scientists had generated, validated and facilitated a large number of hybridomas in the past decade for preclinical research, where the mAb format and isotypes were important to understand their performance in preclinical models. Genetically engineered mAbs are typically produced with recombinant technology, where the variable domains should be sequenced, cloned into plasmids and expressed in transient systems. These processes are time-consuming, challenging and expensive, leading to outsourced work at contract research companies, which hamper the process of academic early-stage antibody development and preclinical research.
In its mechanism of action, the constant antibody domains forming the fragment crystallizable – (Fc) domain are central to the therapeutic efficacy of mAbs since they engage with specific Fc receptors (FcRs). Preceding research work had highlighted the central role of Fc in antibody-based therapeutics to emphasize this role. Since its advent, CRISPR and associated protein Cas-9 (CRISPR-Cas9)-targeted genome editing technology has opened multitudes of exciting opportunities for gene therapy, immunotherapy and bioengineering. Researchers had used CRISPR-Cas9 to modulate mAb expression in hybridomas, generate a hybridoma platform and engineer hybridomas to introduce antibody modification. However, a platform for versatile and effective Fc substitution from foreign species within hybridomas with constant domains remains to be genetically engineered.
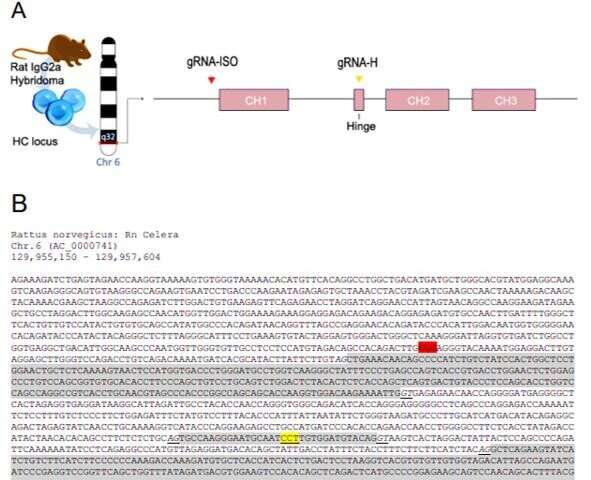
![CRISPR/HDR engineering of hybridoma NLDC145 to obtain sortagable Fab′ fragments against DEC205. (A) Schematic representation of the CRISPR/HDR approach to convert wild-type (WT) hybridomas to Fab′ fragment–producing cell lines. (B) The targeted IgH locus of NLDC-145 with the variable region (VH) and constant regions (CH1, Hinge, CH2, and CH3) annotated is shown. Cas9 is guided by gRNA-H to hinge region and creates a double-stranded break before the first cysteine. The double-stranded break is subsequently repaired by HDR via the donor construct consisting of a sortag and his-tag motif (srt-his), an internal ribosomal entry site (IRES), blasticidin-resistance gene (Bsr), polyA transcription termination signal (pA), and homology arms (5′ HA and 3′ HA). The first 10 amino acids of the hinge before and after successful CRISPR/HDR are indicated. (C) Three days after electroporation, PCR was performed on genomic DNA from WT and CRISPR/HDR-targeted population using primers 1 and 2 (B). Agarose gel shows amplicon of expected size exclusively for the CRISPR/HDR-targeted population, indicating the correct integration of the donor construct within the population. (D) After limiting dilution of CRISPR/HDR-targeted cells, a flow cytometry screen was performed on the supernatant of monoclonal cell suspensions. To this end, DEC205-expressing cells (JAWSII) were incubated with clonal supernatants in combination with secondary antibodies against his-tag (blue) or rIgG2a (red). Exclusive his-tag signal indicates production of Fab′ fragments. (E) Immunoblotting of the supernatant of clones 47 to 52 for his-tag (blue) and rat heavy and light chain (red) confirms flow cytometry results. (F) In competition assay, JAWSII were incubated with a serial dilution (150 to 20 ng/ml) of either purified Fab′DEC205srt (blue), αDEC205 mAb (red), or an isotype control (gray) in combination with fluorescently labeled αDEC205 mAb (1 μg/ml). Decrease in mean fluorescent intensity (MFI) relates to the increase in competition for DEC205 binding with fluorescent-labeled αDEC205 mAb. n = 3, mean ± SEM. (G) Fab′DEC205srt can be C-terminally functionalized with a fluorescently labeled probe [GGGCK(FAM)] by using sortase (3M srt)-mediated ligation. LC, light chain. Credit: Science Advances, doi: 10.1126/sciadv.aaw1822 Genome engineering with CRISPR/HDR to diversify the functions of hybridoma-produced antibodies](https://scx1.b-cdn.net/csz/news/800a/2019/1-genomeengine.jpg)
In the present work, van der Schoot et al. genetically engineered hybridomas using one-step CRISPR/homology-directed repair. They rapidly generated the recombinant hybridomas to secrete mAbs in the Fc format of choice with freedom to install preferred tags or insert mutations. The research team formed hybridomas that produced fragment antigen-binding (Fab') i.e. an antibody site that typically bound to antigens, C-terminally equipped with tags for site-specific chemoenzymatic modification and purification. They generated hybridomas to secrete chimeric antibodies with the isotype and species of choice and form Fc mutant antibodies without compromising antigen specificity. They isolated the engineered antibodies easily from the hybridoma supernatant to observe the expected biochemical and immunological character in vitro and in vivo. Since the research team targeted constant domains of the immunoglobulin locus, the CRISPR/HDR method could be adapted to hybridomas from any species or isotype. The new and versatile method on antibody engineering will empower preclinical antibody research for the scientific community.
Fab' fragments were previously used to treat specific autoimmune diseases, target different therapeutic nanomedicine payloads and generate bispecific antibodies. During conventional technique of obtaining Fab' fragments, scientists could not install useful tags or mutations to the genome. To obtain Fab' secreting hybridomas using CRISPR/HDR in the present work, the research team selected the hybridoma clone NLDC-145 as a first target, which secreted the specific mAbs of interest. To target the heavy chain locus of the clone, van der Schoot et al. used an optimized guide RNA to direct the Cas9 protein to the hinge region of interest and create a double-stranded break. To repair the double-stranded break, they designed an HDR-construct and inserted tags of interest. Since the efficacy of HDR is typically low, the team enriched the media of cells containing the donor construct for competence. They then identified knock-in of the donor construct using the polymerase chain reaction (PCR) to confirm integration of donor constructs into the correct genomic location.
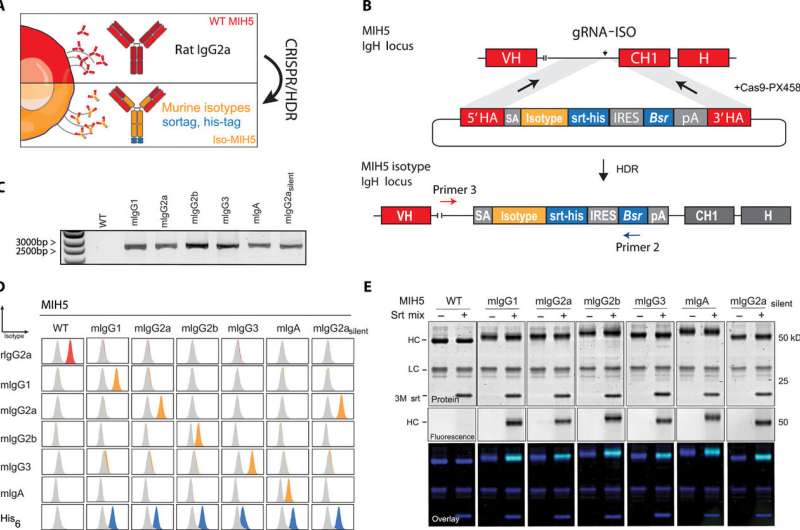
After one week they obtained monoclonal cell suspensions and used the supernatant to determine antigen specificity and phenotype of the secreted product. Using flow cytometry, they observed successful gene engineering of a large fraction of the monoclonal hybridomas and validated the results with western blot analysis to identify the antibody products/proteins of interest. After successfully forming the recombinant hybridomas to produce Fab' of interest, van der Schoot et al. used the same strategy to develop other hybridomas of interest in the field of immuno-oncology. They did not observe a decline in antibody production using the CRISPR/HDR-engineered hybridomas for a long period of time (1 to 3 years).
Encouraged by the success of the one-step CRISPR/HDR strategy to generate Fab' hybridomas, the team continued to expand the platform by engineering hybridomas to secrete diverse Fc variants of the same antibody. As proof-of-concept, they designed a CRISPR/HDR gene trap knock-in approach and tested it by genetically engineering hybridoma MIH5 to produce chimeric antibodies. They included a mutant isotype with amino acid substitutions, which they assessed for on-target, knock-in integration using genomic PCR and verified the presence of the expected PCR product. The research team conducted western blot analysis of the purified chimeric antibody products to confirm substitution of the native rat heavy chain via a genetically introduced mouse heavy chain, at the protein level. They showed that the isotype modification observed using CRISPR/HDR was not merely restricted to the MIH5 hybridomas.
![FcγR engagement of MIH5 isotype variants. Representative sensograms (A) display interactions of MIH5 WT (wild type) and MIH5-engineered mAbs (mIgG1, mIgG2a, mIgG2b, mIgA, and mIgG2asilent) for immobilized mFcγRI, mFcγRIIb, and mFcγRIV at increasing concentrations. Binding to FcγR is expressed in resonance units (RU). SPR was performed on four different concentrations of immobilized FcγRs (1, 3, 10, and 30 nM) to determine affinity [Kd(M)] of the Fc variants for the different FcRs (B). n = 3, mean ± SEM. (C) Predicted antibody-dependent cellular cytotoxicity (ADCC) activity for each murine isotype variant on the basis of their differential affinity for FcγRs. Credit: Science Advances, doi: 10.1126/sciadv.aaw1822 Genome engineering with CRISPR/HDR to diversify the functions of hybridoma-produced antibodies](https://scx1.b-cdn.net/csz/news/800a/2019/3-genomeengine.jpg)
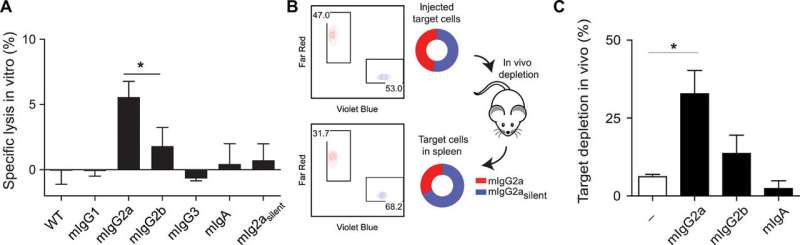
After establishing and validating the hybridoma CRISPR/HDR genomic engineering approach, the team characterized the functional or biological properties of the isotype-switched mAbs. For this, they conducted measurements to determine antibody-receptor interactions. Based on the observed ability to activate and inhibit receptors, van der Schoot et al. predicted the capacity of CRISPR-engineered mAbs to evoke antibody-dependent cellular cytotoxicity—as observed in previous studies. To complete the characterization study, the research team tested the potential of antibodies to induce antibody-dependent cellular toxicity in vitro and in vivo.
For in-lab experiments, they used murine colon adenocarcinoma cells (MC38) opsonized to facilitate phagocytosis/engulfing of the particle, with MIH Fc variants to interact with whole blood. For in vivo experiments, they tested the ability of MIH Fc variants to deplete B cells (that produce immunoglobins). After the experimental procedure, the research team euthanized the mice and isolated their spleens to determine the depletion of B cells using flow cytometry and immunofluorescence imaging. Both in vivo and in vitro assays provided similar results. Together, the work indicated that chimeric and mutant mAbs engineered using CRISPR/HDR technologies exhibited the same biochemical and immune factor characteristics as their native and recombinant counterparts.
In this way, Johan M.S. van der Schoot and co-workers developed a versatile platform for easy and rapid generation of Fc-engineered antibodies, starting from their parent hybridomas. They successfully used a one-step CRISPR/HDR approach to obtain hybridomas, thereby producing (1) monovalent Fab' fragments, (2) Fc isotype variants from a foreign species or (3) Fc-silent mutants without effector functions, within 21 days. The method included useful protein tags for purification and site-specific conjugation. The technique is accessible for any lab capable of hybridoma cell-line culturing.
The process provides a molecular toolbox to repurpose the wealth of established hybridoma cell lines. The universal CRISPR/HDR platform developed in the work had a 100 percent success rate; where each genomic engineering experiment using a single specific HDR construct resulted in multiple correctly engineered hybridoma clones. Since hybridoma products are largely used in preclinical studies in vivo, this method will empower clinical antibody research for therapeutic antibody development. Site-specific functionalization of engineered antibody products will have wide-ranging applications in the fields of biomedical engineering, chemical biology, drug development and nanomedicine, as applicable for the entire scientific community.
More information: Johan M. S. van der Schoot et al. Functional diversification of hybridoma-produced antibodies by CRISPR/HDR genomic engineering, Science Advances (2019). DOI: 10.1126/sciadv.aaw1822
Hélène Kaplon et al. Antibodies to watch in 2018, mAbs (2018). DOI: 10.1080/19420862.2018.1415671
G. KÖHLER et al. Continuous cultures of fused cells secreting antibody of predefined specificity, Nature (2005). DOI: 10.1038/256495a0
M. Jinek et al. A Programmable Dual-RNA-Guided DNA Endonuclease in Adaptive Bacterial Immunity, Science (2012). DOI: 10.1126/science.1225829
Journal information: Science Advances , Nature , Science
Provided by Science X Network
© 2019 Science X Network