A new protein structure that helps viruses with lipid membranes enter cells discovered
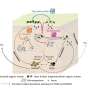
Many viruses, including that perennial winter affliction, the influenza virus, are protected by a lipid membrane on loan from the host cell. The fusion proteins on the surface of the membrane are tasked with merging the lipid membrane of the virus with that of the cell. After this, the virus is able to slip inside the cell, turning it into a virus factory.
The structure and function of the fusion proteins in human and animal viruses have been widely studied, yet the membrane fusion process in unicellular, prokaryotic organisms – archaea and bacteria – remains so far relatively unknown.
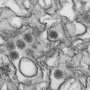
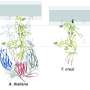
Now, researchers have for the first time determined the atomic structure of a fusion protein of a prokaryotic organism. A fusion protein identified in viruses of prokaryotic archaea, which thrive in hypersaline conditions, turned out to be structurally significantly different from the fusion proteins of human and animal viruses.
"The fusion protein identified in the archaeal virus we studied represents a potentially new, fourth group of proteins," says Elina Roine, senior researcher at the University of Helsinki, Finland.
"This is a significant find, as in the last 40 years, only three types of viral fusion proteins have been found," adds Associate Professor Juha Huiskonen.
Next, the researchers will focus on investigating which fusion proteins needed in the membrane fusion of animal viruses most resemble the viral fusion proteins of this prokaryotic organism. Another topic of interest is whether there are structurally similar proteins among cellular membrane fusion proteins of eukaryotic organisms.
The research project was carried out in collaboration with researchers from Oxford University. The atomic structure of the protein was determined using high-powered X-rays.
More information: Kamel El Omari et al. The structure of a prokaryotic viral envelope protein expands the landscape of membrane fusion proteins, Nature Communications (2019). DOI: 10.1038/s41467-019-08728-7
Journal information: Nature Communications
Provided by University of Helsinki