How bacteria build an enzyme that destroys climate-changing laughing gas

New research from the University of East Anglia reveals how soil bacteria build the only known enzyme for the destruction of the potent global warming and ozone-depleting gas nitrous oxide.
Alongside carbon dioxide (CO2) and methane, the greenhouse gas nitrous oxide (N2O), commonly known as 'laughing gas', is now a cause for great concern, and there is much international focus on reducing emissions.
It is hoped that the findings, published today in the journal Chemical Science, will help pave the way for strategies to mitigate the damaging effects of this climate changing gas.
N2O has around 300 times the global warming potential of CO2 and stays in the atmosphere for about 120 years, where it accounts for around nine per cent of total greenhouse gas.
It also destroys the ozone layer with similar potency to the now banned chlorofluorocarbons (CFCs).
Atmospheric levels of N2O are rising year on year as microorganisms break down synthetic nitrogen fertilisers which are added to agricultural soil, to satisfy the food supply demands of an ever-increasing global population.
Prof Nick Le Brun from UEA's School of Chemistry, said: "It is well known that some bacteria can 'breathe' N2O in environments where oxygen (O2) is limited.
"This ability is entirely dependent on an enzyme called 'nitrous oxide reductase', which is the only enzyme known to destroy N2O. It is therefore very important for controlling levels of this climate-changing gas.
"We wanted to find out more about how soil bacteria use this enzyme to destroy nitrous oxide."
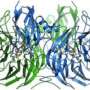
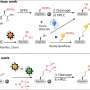
The part of the enzyme where N2O is consumed (called the 'active site') is unique in biology, consisting of a complex arrangement of copper and sulfur (a copper-sulfide cluster). Until now, knowledge of how this unusual active site is built by bacteria has been lacking.
The UEA team discovered a protein called NosL, which is required for the assembly of the copper-sulfide cluster active site and makes the enzyme active.
They found that bacteria lacking NosL still produced the enzyme but it contained less of the copper-sulfide active site. Furthermore, when the same bacteria were grown with copper in short supply, the active site was completely absent from the enzyme.
The team also showed that NosL is a copper-binding protein, indicating that it functions directly in supplying copper for the assembly of the copper-sulfide cluster active site.
Prof Le Brun said: "The discovery of the function of NosL is the first step towards understanding how the unique active site of nitrous oxide reductase is assembled. This is key information because when assembly goes wrong, inactive enzyme leads to release of N2O into the atmosphere."
The UEA team was led by Prof Nick Le Brun and Dr. Andy Gates from UEA's School of Biological Sciences, and included the University's Vice Chancellor Prof David Richardson—also from the School of Biological Sciences. They are part of international EU network focussed on understanding different aspects of N2O and the nitrogen cycle.
Dr. Gates said: "Society is generally well aware of the need to address carbon dioxide emissions, but nitrous oxide is now emerging as a pressing global concern and requires researchers with different skill sets to work together to prevent further damaging effects of climate change.
"With increasing understanding of the enzymes that make and destroy N2O, we move closer to being able to develop strategies to mitigate the damaging effects of this climate changing gas on the earth's environment."
More information: 'NosL is a dedicated copper chaperone for assembly of the Cuz center of nitrous oxide reductase' is published in Chemical Science, a peer-reviewed journal published by the Royal Society of Chemistry, on April 18, 2019. pubs.rsc.org/en/content/articl … c01053j#!divAbstract
Journal information: Chemical Science
Provided by University of East Anglia