New magnetically controlled thrombolytic successfully passed preclinical testing

A new anti-thrombosis drug based on magnetite nanoparticles developed at ITMO University has been successfully tested on animals. Preclinical studies showed the drug's high efficacy and lack of side effects. Clot dissolution time of the new drug is 20 times shorter than traditional medications. The range of permissible concentrations is very high, and the minimum dose of the active substance required to achieve the effect was 100 times smaller than usual. The results are published in Applied Materials and Interfaces.
Currently, thrombosis-related conditions remain the leading cause of death. There are two conventional ways of treatment: surgery, requiring a complex, high-risk operation, or thrombolytics. Although this class of drugs appeared as a treatment method about 40 years ago, it is not widespread because of the side effects that occur during systemic use. To avoid these effects, the action of thrombolytics should be localized, meaning that the drug must be delivered right to the clot. In order to do this, scientists use magnetic nanoparticles.
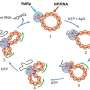
Researchers at ITMO University developed thrombolytics based on magnetite nanoparticles coated with heparin and urokinase. Magnetite is a biocompatible iron oxide with pronounced magnetic properties. Therefore, magnetite particle movement can be controlled by a magnetic field. Urokinase is a first-generation thrombolytic with simple molecules, affordability and efficiency almost equal to newer drugs. As soon as the nanoparticles with urokinase are injected into the blood, they can be sent to the site of clot formation using a magnetic field. Once the clot is destroyed, the magnetic field is turned off, and the nanoparticles are redistributed to the liver and spleen and, finally removed.
"We initially focused on simple and inexpensive substances to make the final product affordable. Since urokinase and magnetite are equally charged, we had to use a linker. We choose heparin, an anticoagulant that often comes with thrombolytics in order to thin the blood. Typically, heparin inhibits urokinase, but we managed to avoid this effect. Preclinical trials showed that we also managed to achieve high efficiency and minimize side effects," comments Arthur Prilepskii, member of SCAMT Laboratory of ITMO University.
The new drug successfully passed preclinical studies including toxicity, allergenicity, mutagenicity and immunotoxicity tests. No side effects were identified during animal experiments. The range of permissible drug concentrations turned out to be very high, while the minimum dose of urokinase, necessary to achieve a therapeutic effect, was approximately two orders of magnitude lower than with the introduction of the usual urokinase. Moreover, the clot dissolution time was 20 times shorter.
"Preclinical trials were conducted as part of the Pharma 2020 project. The project included 3 stages for 2 years, during which the synthesis of the drug was optimized. Moreover, we carefully studied chemical characteristics efficacy and safety of the new medication," notes Anna Fakhardo, researcher at SCAMT Laboratory of ITMO University.
More information: Artur Y. Prilepskii et al, Urokinase-Conjugated Magnetite Nanoparticles as a Promising Drug Delivery System for Targeted Thrombolysis: Synthesis and Preclinical Evaluation, ACS Applied Materials & Interfaces (2018). DOI: 10.1021/acsami.8b14790
Journal information: ACS Applied Materials and Interfaces
Provided by ITMO University