Producing defectless metal crystals of unprecedented size
A research group at the Center for Multidimensional Carbon Materials, within the Institute for Basic Science (IBS), has published an article in Science describing a new method to convert inexpensive polycrystalline metal foils to single crystals with superior properties. These materials have many applications in science and technology.
The structure of most metal materials can be thought of as a patchwork of tiny crystals bearing some defects on the borders between each patch. These defects, known as grain boundaries (GBs), worsen the electrical and sometimes mechanical properties of the metal. By contrast, single crystal metals have no GBs and show higher electrical conductivity and other enhanced qualities that can play a major role in fields such as electronics, plasmonics and catalysis, among others. Single crystal metal foils have also attracted much attention because certain single crystal metals, such as copper, nickel and cobalt are suitable substrates for the growth of defectless graphene, boron nitride and diamond.
Single crystals are normally fabricated beginning with a so-called "crystal seed." Conventional approaches, such as the Czochralski or Bridgman methods, or others based on the deposition of thin metal films on single crystal inorganic substrates, achieve small single crystals at high processing costs.
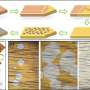
To unlock the full potential of such metal structures, the IBS team led by Rodney Ruoff at Ulsan National Institute of Science and Technology (UNIST), along with Jin Sunghwan and Shin Hyung-Joon, invented the contact-free annealing (CFA) technique. CFA involves heating the polycrystalline metal foils to a temperature slightly below the melting point of each metal. This new method does not need single crystal seeds or templates that limit the maximum crystal size. The method was tested with five different types of metal foils: copper, nickel, cobalt, platinum and palladium. It resulted in a colossal grain growth, reaching up to 32 square centimeters for copper.
The details of the experiment varied according to the metal used. In the case of copper, the researchers used quartz holders and a rod to hang the metal foil like clothes suspended on clotheslines. Then the foil was heated for several hours in an atmosphere of hydrogen and argon in a tube-shaped furnace to approximately 1050 degrees Celsius (1323 degrees Kelvin), a temperature close to copper's melting point (1358 K), and then cooled down.
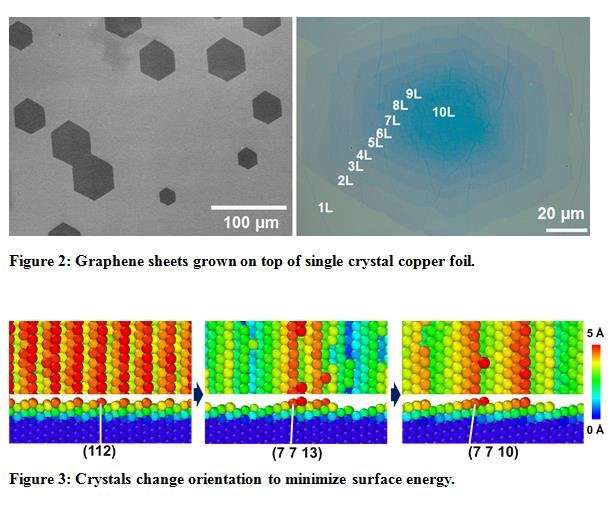
Figure 3: Crystals change orientation to minimize surface energy. Members of the team, DING Feng, ZHANG Leining, and DONG Jichen, contributed a model and theoretical calculations on 'colossal grain growth' that was observed and studied through experiments. SHIN Hyung-Joon drew attention to the importance of the initial "texture" of the polycrystalline metal foils and the role this plays in colossal grain growth. In the presence of large amounts of vacancies within the crystal structure (5 percent in these molecular dynamics simulation), the crystal orientation changes to minimize the surface energy. Credit: IBS
The scientists also achieved single crystals from nickel and cobalt foils, each about 11 cm2. The achieved sizes are limited by the size of the furnace. For platinum, resistive heating was used because of its higher melting temperature (2041 K). Current was passed through a platinum foil attached to two opposing electrodes, then one electrode was moved and adjusted to keep the foil flat during expansion and contraction. The research team expects this trick to work for other foils, because it also worked for palladium.
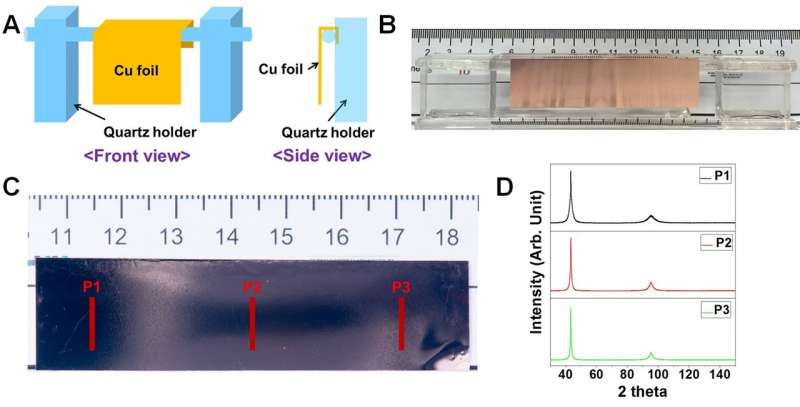
These large single crystal metal foils are useful in several applications. For example, they can serve to grow graphene on top of them. The group obtained high-quality single crystal monolayer graphene on single crystal copper foil, and multilayer graphene on a single crystal copper-nickel alloy foil.
The new single crystal copper foil showed improved electrical properties. Collaborators Yoo Won Jong and Moon Inyong at Sungkyunkwan University measured a 7 percent increase in the room temperature electrical conductivity of the single crystal copper foil, compared to the commercially available polycrystalline foil.
"Now that we have explored these five metals and invented a straightforward scalable method to make such large single crystals, there's the exciting question of whether other types of polycrystalline metal films, such as iron, can also be converted to single crystals," notes first author of the study, Jin Sunghwan.
Ruoff, his supervisor, says, "Now that these cheap single crystal metal foils are available, it will be tremendously exciting to see how they are used by the scientific and engineering communities."
More information: Science (2018). DOI: 10.1126/science.aao3373
Journal information: Science
Provided by Institute for Basic Science