Synthesis of the veterinary antibiotic florfenicol by a fast chemo-enzymatic route
Fluorinated structures make up more than 20 percent of modern drugs, but benign and fast fluorination schemes are scarce. In a study published in the European Journal of Organic Chemistry, scientists have now combined dynamic kinetic resolution and nucleophilic fluorination for the asymmetric synthesis of florfenicol, a veterinary antibiotic. After enzymatic resolution of the racemate, two different fluorination strategies both proved effective and fast. One is especially interesting for industrial production.
Fluorine substituents have many welcomed effects in drugs. Often, fluorinated compounds are more lipophilic, penetrate more easily in the cells, and enzymes may find it harder to degrade them. For example, the fluorinated antibiotic florfenicol is more active than thiamphenicol, to which it is related by a terminal fluorine instead of a hydroxy group. Florfenicol and thiamphenicol, which are both variants of chloramphenicol, are used against conjunctivitis and respiratory diseases in cattle.
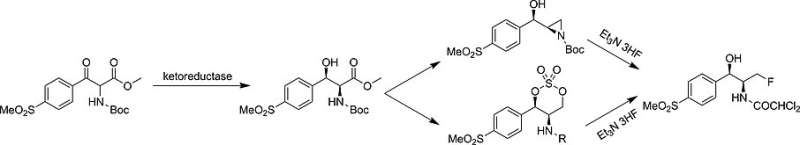
Despite the relevance of fluorination, many industrial fluorination strategies still involve aggressive and harmful reagents. In the case of florfenicol, not only is the fluorinating agent expensive and corrosive, but the preparation of the two stereocenters also produces a high amount of waste. These issues prompted Fuli Zhang from China State Institute of Pharmaceutical Industry, Shanghai, China, and his colleagues, to look for more environmentally benign processes. To synthesize florfenicol, they had to construct two adjacent stereocenters of the cis-1,2-amino alcohol and introduce a terminal fluorine atom.
The scientists chose to install the fluorine as a last, separate step because the two asymmetric carbon atoms can be conveniently prepared by dynamic kinetic resolution. Kinetic resolution means that two enantiomers can be separated according to their different reactivity. The process is called "dynamic" when the two enantiomers can racemize, that is, interconvert. Then, the faster reacting enantiomer can be isolated with maximal yield.
Zhang and his colleagues designed a dynamic reductive kinetic resolution using ketoreductase enzymes as biocatalysts and glucose as the hydride source. Having established the two stereocenters by this bioenzymatic route, they explored possible fluorination reactions. Two routes of nucleophilic fluorination proved especially convincing, and both routes use amine hydrofluoride as the fluorinating reagent, which is mild and selective and can be handled in glass equipment.
For industrial manufacture, the authors also have a favorite. The route involving a cyclic sulfate intermediate outcompeted the other one containing an intermediate aziridine, a three-membered nitrogen-containing ring. The chemo-enzymatic sulfate route produced florfenicol in only five steps and showed high yield and flexibility as well as acceptable environmental and health safety. The next step will be the further exploration of the reductase enzymes in other dynamic bioreduction syntheses, the authors remark.
More information: Jie Zou et al. Asymmetric Synthesis of Florfenicol by Dynamic Reductive Kinetic Resolution with Ketoreductases, European Journal of Organic Chemistry (2018). DOI: 10.1002/ejoc.201800658
Journal information: European Journal of Organic Chemistry
Provided by Wiley