Superconductivity above 10 K in a novel quasi-one-dimensional compound
In the past century, superconductivity has been observed in thousands of substances with multifarious chemical compositions and crystal structures; however, researchers have still not found an explicit method for discovering new superconductors. For the unconventional high-Tc superconductors of cuprates and iron pnictides/chalcogenides, the occurrence of superconductivity is highly related to the existence of some certain quasi-two-dimensional structural motifs, e.g., the CuO2 planes or the Fe2As2/Fe2Se2 layers. Thus, low dimensionality has generally been considered as a favorable ingredient for exotic electron pairing due to the enhancement of electronic correlations. While among the quasi-one-dimensional (Q1D) compounds, only a few compounds were found to be superconducting at considerably low temperatures of several degrees Kelvin.
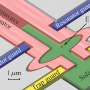
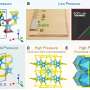
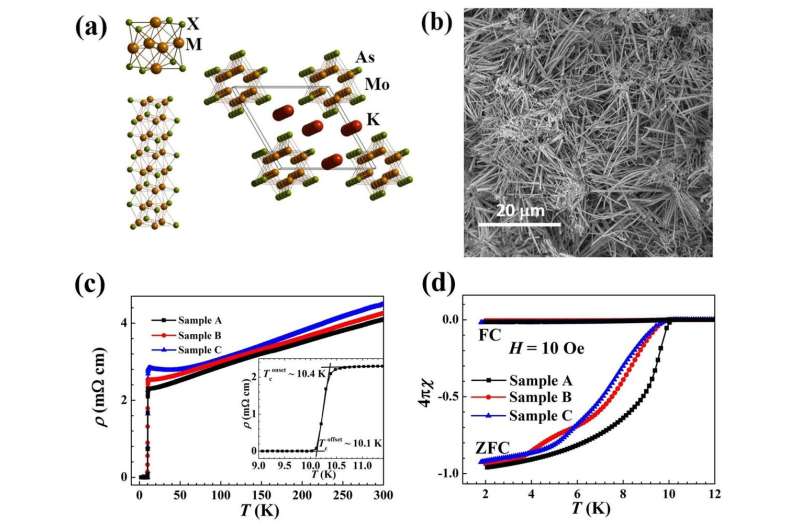
Recently, a team led by Prof. Zhian Ren from the Institute of Physics, Chinese Academy of Sciences discovered a Q1D superconductor K2Mo3As3, with a Tc value exceeding 10 K for the first time. Although many molybdenum chalcogenide superconductors were discovered in the 1970s, ternary compounds of molybdenum arsenide have rarely been reported. After much study of Mo-based ternary phases, the team succeeded in synthesizing the new K2Mo3As3 compound, which crystalizes in a noncentrosymmetric hexagonal structure with typical Q1D (Mo3As3)2- linear chains separated by K+ cations, similar to the structure of K2Mo3As3. Bulk superconductivity below 10.4 K was confirmed by electrical resistivity, magnetic susceptibility, and heat capacity measurements. The K2Mo3As3 is the first MoAs-based superconductor and possesses the record Tc in all Q1D superconductors. This discovery indicates that Cr and Mo based Q1D superconductors may share some common underlying origins within the similar structural motifs and will help to uncover the exotic superconducting mechanism in low dimensional materials.
More information: Qing-Ge Mu et al, Superconductivity at 10.4 K in a novel quasi-one-dimensional ternary molybdenum pnictide K 2 Mo 3 As 3, Science Bulletin (2018). DOI: 10.1016/j.scib.2018.06.011
Provided by Science China Press