High-performance multimetallic core-interlayer-shell icosahedral electrocatalysts for ORR
Proton-exchange membrane fuel cells (PEMFCs) are generally regarded as a clean and sustainable energy-conversion technology to replace increasingly scarce fossil fuels due to the high energy conversion efficiency, high energy density, and low or zero pollutant emission. Clearly, platinum (Pt) is a key component of the state-of-the-art electrocatalysts for the oxygen reduction reaction (ORR) at the cathodes by far, which is a determining half reaction to boost the performance of this fuel cell technology. However, the sluggish reaction kinetics of ORR often requires a relatively high load of Pt to achieve a desirable fuel cell performance in the practical applications, which is severely restricted by the high cost and scarcity of Pt.
As such, researchers have made tremendous efforts to develop active, stable, and economical Pt-based catalysts towards ORR over the past decade. Incorporating Pt with less noble and/or low-cost metals to form alloy/core-shell catalysts is regarded as a promising strategy to substantially enhance the catalytic properties towards ORR in fuel cells due to electron coupling and strain effect between distinct metals. Compared with Pt-based alloy electrocatalysts, such core-shell electrocatalysts not only avoid the leaching or dissolution of the non-Pt metal to improve stability, but also make full use of every Pt atom and thus cut down the cost of catalysts.
According to the well-known volcano plot for ORR activity, Pd is probably the best core material to form Pt-based core-shell electrocatalysts due to the suitable electron coupling and synergetic effect between them. However, the stability of such Pd@Pt core-shell catalysts cannot meet the requirement of the commercialization due to the selective dissolution of Pd atoms from the cores during the cycling. The addition of stabilizing elements (e.g., Au) into Pt-based nanocrystals has proved to protect active components (e.g., Pt and Pd) in the catalysts from dissolution upon potential cycling by up-shifting their dissolution potential, and thereby guarantees excellent long-term stability. However, these Au@Pt core-shell electrocatalysts suffer from decrease in ORR activity due to the induced expansive strain in Pt shells arising from the larger lattice constant and bigger atomic size of Au than Pt in combination with the adverse electronic coupling between them.
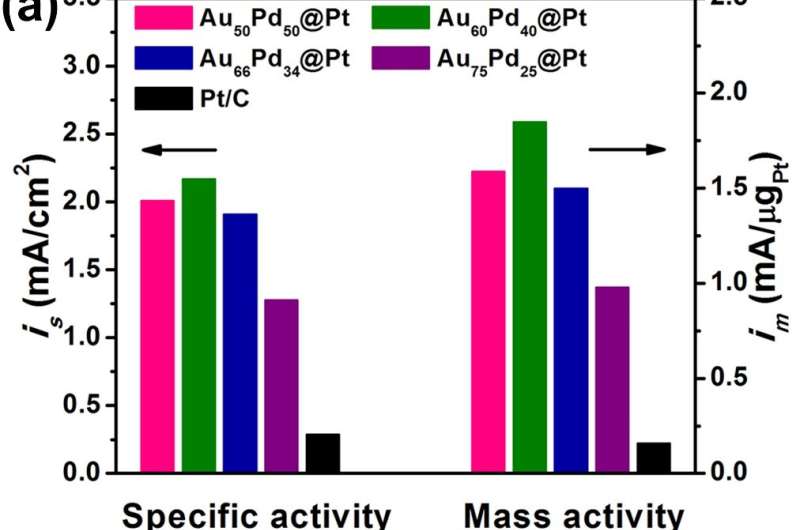
Recently, Prof. Zhang in the research team led by Prof. Deren Yang from Zhejiang University collaborated with Prof. Wu from Shanghai Jiao Tong University developed a facile approach to synthesize icosahedral AuPd@Pt nanocatalysts consisting of ultrathin Pt skins, Au-Pd alloy cores and Pd interlayers with variable thickness. Ultrathin Pt skins with two atomic layers were epitaxially coated on as-prepared icosahedral Au-Pd seeds. The control on the thickness of Pd interlayers in a range of 3~12 atomic layers was realized by tuning the feeding molar ratio of Au and Pd elements. The AuPd@Pd@Pt icosahedra with Pd interlayers, especially Au60Pd40@Pt electrocatalysts with about six atomic layers, exhibited remarkably enhanced activities and durabilities towards ORR in acid environment compared with commercial Pt/C and Au75Pd25@Pt icosahedra without Pd interlayers. The three-layered core-interlayer-shell nanostructures with icosahedral shapes further optimize the electronic structure of noble metal atoms for ORR catalysis and maximize the utilization of Pt on the basis of other core-shell nanocatalysts. This work shed light on the key roles of Pd interlayers in high-performance core-shell Au-Pd-Pt ORR catalysts and provides a new strategy for electrocatalysts design, which ensures high activity and durability simultaneously.
More information: Qingfeng Xu et al, Multimetallic AuPd@Pd@Pt core-interlayer-shell icosahedral electrocatalysts for highly efficient oxygen reduction reaction, Science Bulletin (2018). DOI: 10.1016/j.scib.2018.03.013
Provided by Science China Press