Chemical compound produces beneficial inflammation, remyelination that could help treat MS
Drugs available to treat multiple sclerosis, a leading cause of neurological disability affecting roughly 2.3 million people worldwide, alter the body's immune system to reduce disease symptoms and disability.
They do not induce, however, repair of damaged axons, the long threadlike parts of nerve cells that conduct impulses between cells or restore myelin, the protective sheath that surrounds the axons of neurons essential for the proper functioning of the brain and spinal cord.
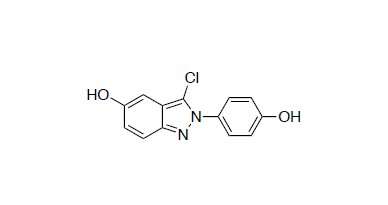
Researchers at the University of California, Riverside, now report that indazole chloride, a synthetic compound that acts on one form of the body's estrogen receptors previously shown to reduce multiple sclerosis symptoms in mouse models, is able to do both: remyelinate (add new myelin to) damaged axons and alter the immune system.
"While additional translational studies are required, indazole chloride and similar drugs may represent a promising new avenue of treating the underlying loss of myelin in multiple sclerosis," said Seema Tiwari-Woodruff, an associate professor of biomedical sciences in the School of Medicine, who led the mouse study.
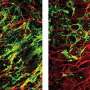
Multiple sclerosis is triggered when the immune system attacks and damages the myelin sheath. As myelin is lost, nerve signals slow down or stop, affecting the patient's vision, movement, memory, and more. Oligodendrocytes are the mylenating cells of the central nervous system. Normally, oligodendrocyte precursor cells mature into myelin-producing oligodendrocytes when myelin is damaged.
This process often fails, however, in multiple sclerosis, resulting in permanent damage. The UCR researchers found the change in the immune system provides a protective shield for oligodendrocytes, preventing this damage and possibly even reversing it.
"With remyelination of axons, nerve impulses travel faster than before, thus decreasing multiple sclerosis disability," Tiwari-Woodruff said. "As a potential therapy for the treatment of multiple sclerosis, indazole chloride may represent the first in a novel class of drugs capable of reducing disability burden in patients with multiple sclerosis. We still don't know the mechanism of action of pre-clinical therapies like indazole chloride. Our report aims to understand how drugs like indazole chloride are working so we can make more selective and efficacious drugs."
Study results appear this week in the Proceedings of the National Academy of Sciences.
Indazole chloride, a ligand, stimulates ERβ, an estrogen receptor in the body. Indazole chloride is an attractive drug because it does not produce the negative side-effects of estrogen therapy. Because ERβ are present not just in oligodendrocytes but also in microglia, neurons, and T-cells, indazole chloride may have therapeutic benefits not just for multiple sclerosis, but also other autoimmune diseases.

Tiwari-Woodruff explained that while inflammation causes a lot of damage in autoimmune diseases, not all inflammation is harmful. Beneficial inflammation is required to fight infectious disease and speeds up wound healing by clearing dead cells and tissue. Indazole chloride reduces "bad" inflammation and promotes "good" inflammation, thereby protecting new oligodendrocytes while they remyelinate. Tiwari-Woodruff and her group found that indazole chloride accomplishes this by strengthening the production of a molecule called "CXCL1," which makes oligodendrocytes resistant to "bad" inflammatory signals.
Tiwari-Woodruff's findings provide a stepping stone toward a way to repair the damage to axons and oligodendrocytes caused by multiple sclerosis. In collaboration with John Katzenellenbogen, a research professor of chemistry at the University of Illinois at Urbana-Champaign, Tiwari-Woodruff's group is screening chemically similar analogs of indazole chloride for more efficacious and safe therapy. Provisional patents have been filed for some of these analogs.
"It's quite possible we may find an analog far superior to indazole chloride," Tiwari-Woodruff said.
More information: Hawra Karim et al. Increase in chemokine CXCL1 by ERβ ligand treatment is a key mediator in promoting axon myelination, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1721732115
Journal information: Proceedings of the National Academy of Sciences
Provided by University of California - Riverside