Rabies trick could help treat Parkinson's disease
The rabies virus wreaks havoc on the brain, triggering psychosis and death. To get where it needs to go, the virus must first trick the nervous system and cross the blood brain barrier—a process that makes it of interest in drug design. Now, scientists report in ACS Nano a way to exploit the rabies virus machinery to deliver a Parkinson's disease medication directly to the brain.
Parkinson's disease, the slow degeneration of the brain cells that control movement, affects about a million Americans, according to the Parkinson's Foundation, and has no cure. While the exact cause of Parkinson's disease is unknown, a common feature of the illness is the accumulation of iron in neurons, inflicting damage and cell death. Some doctors are now using a metal-grabbing compound called deferoxamine to sop up the excess iron in patients, but high doses are needed due to the drug's limited capacity to enter the brain, bringing on serious side effects. To lower the effective dose, Yan-Zhong Chang, Xin Lou, Guangjun Nie, and colleagues wanted to take advantage of a key part of the rabies virus to usher deferoxamine into the brain.
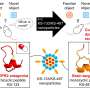
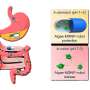
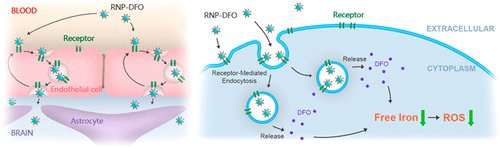
Glycoprotein 29 is a part of the rabies virus that binds to a brain cell receptor and crosses the blood brain barrier. The researchers attached glycoprotein 29 to a nanoparticle stuffed full of deferoxamine. Then, they injected the iron-grabbing nanoparticles into mouse models of Parkinson's disease. The iron levels in the mouse brains dropped, reducing the brain damage and reversing the disease symptoms, without noticeable side effects. Since all of the components in the therapeutic agent are already approved for use in the clinic, the researchers are looking toward human trials.
More information: Linhao You et al. Targeted Brain Delivery of Rabies Virus Glycoprotein 29-Modified Deferoxamine-Loaded Nanoparticles Reverses Functional Deficits in Parkinsonian Mice, ACS Nano (2018). DOI: 10.1021/acsnano.7b08172
Abstract
Excess iron deposition in the brain often causes oxidative stress-related damage and necrosis of dopaminergic neurons in the substantia nigra and has been reported to be one of the major vulnerability factors in Parkinson's disease (PD). Iron chelation therapy using deferoxamine (DFO) may inhibit this nigrostriatal degeneration and prevent the progress of PD. However, DFO shows very short half-life in vivo and hardly penetrates the blood brain barrier (BBB). Hence, it is of great interest to develop DFO formulations for safe and efficient intracerebral drug delivery. Herein, we report a polymeric nanoparticle system modified with brain-targeting peptide rabies virus glycoprotein (RVG) 29 that can intracerebrally deliver DFO. The nanoparticle system penetrates the BBB possibly through specific receptor-mediated endocytosis triggered by the RVG29 peptide. Administration of these nanoparticles significantly decreased iron content and oxidative stress levels in the substantia nigra and striatum of PD mice and effectively reduced their dopaminergic neuron damage and as reversed their neurobehavioral deficits, without causing any overt adverse effects in the brain or other organs. This DFO-based nanoformulation holds great promise for delivery of DFO into the brain and for realizing iron chelation therapy in PD treatment.
Journal information: ACS Nano
Provided by American Chemical Society