Genes' interplay gives clues to how new cell types could evolve
Developmental biologists at the University of Bath have gained insights into how a family of essential genes interact differently between different parts of the body and between species, which could offer clues about how new types of cells come to evolve.
The researchers from the Department of Biology & Biochemistry, working with colleagues from two Japanese universities, were interested in how an important family of regulatory genes called SOX genes affect cell development as zebrafish and medaka fish grow.
SOX genes are found in all animals, such as mammals (including humans), reptiles, birds, fish and insects , and are known to be crucial in the development of brain cells, stem cells and in various other types of specialised cell as they develop from their precursor cells.
There are around 20 SOX genes which code for proteins called SOX transcription factors. These act to regulate when genes are switched on and off by attaching to DNA strands, controlling activity of the genes nearby.
Uncovering how these genes function has important consequences for understanding how the body makes different cell-types, and for diseases in which this fails. For example, SOX10 has long been associated with diseases in which melanocytes and neurons fail to be made, such as Waardenburg Syndrome and Hirschsprung Disease.
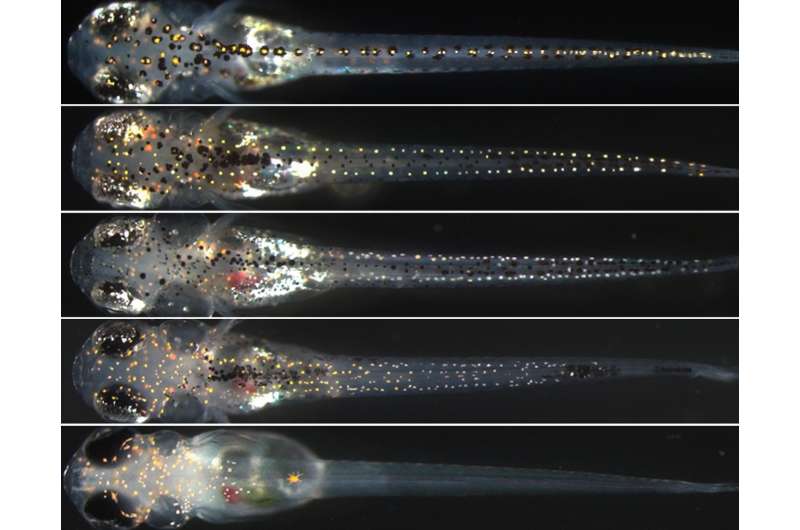
The team were interested in how the SOX proteins interacted when pigment cells develop in these two fish species. Zebrafish have three types of pigment cells called melanocytes, iridophores and xanthophores, while Medaka have the same three and an additional type called leucophores; together these cells form the important and often beautiful pigment patterns of these and other fish.
They studied two SOX proteins, encoded by the SOX10 and SOX5 genes, which are involved in pigment cell development in both species.
They discovered that across both species SOX10 is essential to develop the three types of pigment cells they have in common, and SOX5 slightly downregulated the action of SOX10 for all three types of pigment cell in zebrafish, and for melanocytes and iridophores in medaka.
However in medaka fish both SOX10 and SOX5 worked in a completely different way by co-operating to repress leucophore formation, and to promote the formation of xanthophores.
The research is published in PLOS Genetics.
Professor Robert Kelsh, who led the research at the University of Bath, said: "How individual cells become specific cell-types, from precursor cells that could become anything, is a fundamental question in developmental and stem cell biology.
"We have worked on SOX10 for a long-time, showing its important role in helping make many specialised cell-types. We began looking at the role for SOX5 because this gene has been shown to work with SOX10, but in different ways. Our research shows that these transcription factors are working in a context-specific way - in some cell-types they work together, in others SOX5 antagonises SOX10. For melanocytes this antagonism functions the same way as it does in mammals. But what really surprised us was how SOX5 and SOX10 interact in opposite ways to govern development of xanthophores.
"We see the same proteins are working together differently depending on the context - so what's happening? We suggest that this likely relates to the evolution of a novel pigment cell type - the reflective leucophore - in medaka. Our previous work had shown that at a genetic level we can consider the leucophore to be a xanthophore that has been modified to become reflective. We speculate that this may be intimately linked to the change in relationship between SOX5 and SOX10 in the formation of the xanthophore. But exactly why, and exactly how this works, we don't know - that is the next mystery for us to investigate!"
More information: Distinct interactions of Sox5 and Sox10 in fate specification of pigment cells in medaka and zebrafish, PLOS Genetics (2018). journals.plos.org/plosgenetics … journal.pgen.1007260
Journal information: PLoS Genetics
Provided by University of Bath