Researchers produce 50x more stable adsorbent
A KAIST research team have developed a technology to increase the stability of amine-containing adsorbents by 50 times, moving another step toward commercializing stable adsorbents that last longer.
Professor Minkee Choi from the Department of Chemical and Biomolecular Engineering and his team succeeded in developing amine-containing adsorbents that show high oxidative stability.
The capture of the greenhouse gas carbon dioxide is an active ongoing research field, and some of the latest advancements point to amine-containing adsorbents as an efficient and environmentally friendly way to capture carbon dioxide. However, existing amine-containing adsorbents are known to be unstable under oxidation, which chemically breaks down the adsorbent, thereby making it difficult to rely on amine-containing adsorbents for repeated and continued use.
The researchers have discovered that the miniscule amount of iron and copper present in the amine accelerate the oxidative breakdown of the amine-containing adsorbent. Upon this discovery, they proposed the use of a chelator substance, which essentially suppresses the activation of the impurities.
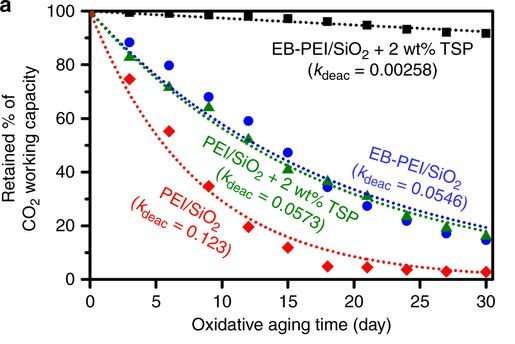
The team demonstrated that the proposed method renders the adsorbent up to 50 times more slowly in its deactivation rate due to oxidation, compared to conventional polyethyleneimine (PEI) / silica adsorbents. Figure 1 illustrates the superior performance of this oxidation-stable amine-containing adsorbent (shown in black squares), whose carbon dioxide-capturing capacity deteriorates by only a small amount (~8 percent). Meanwhile, the carbon dioxide-capturing capacity of the PEI/silica adsorbent (shown in red diamonds) degrades dramatically after being exposed to oxidative aging for 30 days.
This stability under oxidation is expected to bring amine-containing adsorbents one step closer to commercialization. Professor Choi says that commercialization steps for his team's carbon dioxide adsorbents are already underway. He further aims to "develop the world's best carbon dioxide capture adsorbent."
More information: Kyungmin Min et al, Oxidation-stable amine-containing adsorbents for carbon dioxide capture, Nature Communications (2018). DOI: 10.1038/s41467-018-03123-0
Journal information: Nature Communications




















