Phosphate rock an effective fertilizer in Kenya

Farming in western Kenya is challenging, to put it mildly. Although farmers can cycle two full crops in a single year, extremely poor soils and expensive traditional fertilizers, such as triple superphosphate (TSP), keep yields low. But results of a new University of Illinois study offer Kenyan farmers hope in the form of phosphate rock.
"Phosphate rock matches or slightly outperforms triple superphosphate if you're looking at traditional agronomic assessments of soil phosphorus. But if you look at the benefits in terms of soil biology and health, phosphate rock skyrockets ahead of TSP," says Andrew Margenot, assistant professor in the Department of Crop Sciences at U of I and lead author on the study.
Margenot and his team, which includes researchers from the University of California, Davis and the International Center for Tropical Agriculture (CIAT) in Nairobi, investigated the two phosphorus fertilizers in a long-term field trial on a typical Kenyan smallholder farm.
Triple superphosphate, which is about 45 percent phosphorus, must be imported from Moroccan mines. The fertilizer is highly soluble in the acidic, iron-rich soils typical of the region, but it presents several problems.
"It's often not possible for farmers to afford the amount of phosphorus that's recommended if you buy it in the form of imported TSP," Margenot explains. "Plus, the iron in these soils combined with the low pH will bind up and immobilize the phosphorus in TSP. So you just wasted a pretty expensive investment."
Phosphate rock, a fertilizer with only 8 to 12 percent phosphorus, is not only inexpensive, it is naturally suited for use in acidic soils.
"It's the same mineral that our teeth and bones are made out of: calcium phosphate. For the same reason that acidic soda degrades your teeth, phosphate rock will dissolve best and be most useful as a fertilizer in low pH soils," Margenot says.
In the study, the researchers applied TSP, phosphate rock, or no additional fertilizer to fields planted in a corn-bean rotation for a total of 13 cropping seasons. At the conclusion of the study, the team collected soil and measured crop-available phosphorus and the amount that was bound to iron.
In addition, the team also measured the amount of phosphorus that ended up in the soil microbial community. "The thinking is that microbes are able to outcompete geochemical sinks for phosphorus," Margenot says. "The microbe can dart in and siphon up the pulse of phosphorus before it's fixed forever by the iron. As microbial cells die, they free up their phosphorus, giving plant roots an opportunity to take it up."
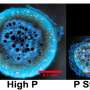
Traditional measures of crop-available phosphorus were similar for TSP and phosphate rock, but about 33 percent more phosphorus was bound to iron, and thus unavailable to crops, in TSP-fertilized plots. Impressively, 299 percent more phosphorus was present in the microbial pool in soils fertilized with phosphate rock.
"Phosphate rock is a form of slow-release fertilizer. TSP is a flood," Margenot says. "There's too much at once, it's like trying to drink from a firehose. It's too much for the microbes or the plant to take up, and so most of it just gets locked up by soil minerals forever."
Yield effects of the fertilizers were reported in a companion study, which indicated that phosphate rock matched and, in some cases, outperformed TSP. Margenot says that's partially due to the greater availability of phosphorus, but it's also due to the fact that phosphate rock lowers acidity and provides additional nutrients – magnesium and calcium – to the soil and, ultimately, crops.
The article, "Biological P cycling is influenced by the form of P fertilizer in an Oxisol," is published in Biology and Fertility of Soils.
More information: Andrew J. Margenot et al. Biological P cycling is influenced by the form of P fertilizer in an Oxisol, Biology and Fertility of Soils (2017). DOI: 10.1007/s00374-017-1226-9
Provided by University of Illinois at Urbana-Champaign