Pearly material for bendable heating elements
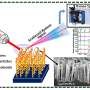
The iridescent shimmer of a string of pearls may one day be more than pretty adornment. Scientists now report in ACS Applied Nano Materials a hybrid material consisting of imitation pearl combined with silver nanowires that works as a heater, with the added benefit of high flexibility, suggesting a potential role in wearable devices.
Nacre, otherwise known as mother of pearl, makes up the opalescent exterior coating of a pearl and the inner coating of mollusk shells. Scientists have long been interested in this lightweight, yet strong material for a range of applications, manufacturing substances that imitate nacre for use as fire retardants, gas barriers and conductors. Flexible conductors made from silver nanowires and a flexible plastic substrate called polyethylene terephthalate (PET) are being developed in laboratories, but researchers are running into problems with adhesion between PET and the silver nanowires. So, Hongwei Duan and colleagues wanted to explore a nacre mimetic as the substrate for silver nanowires to develop a flexible resistive heater.
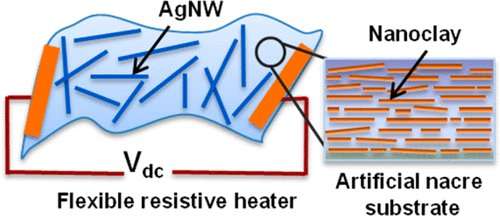
The researchers prepared the nacre mimetic from a synthetic nanoclay coated with a polyvinyl alcohol polymer that self-assembled upon water removal, generating a flexible transparent sheet. Then, they transferred silver nanowires from a filter membrane to the nacre mimetic sheet by hot pressing the pearly substrate against the membrane. The silver nanowires adhered to the new material better than to traditional flexible plastic substrates, resisting removal with scotch tape, a standard test of adhesion. The composite material showed excellent heating properties, producing heat as electric current was passed through the silver nanowires, even when bent repeatedly or when rolled up into a tube. In a proof-of-concept experiment, the researchers placed a heat-sensitive antibiotic-loaded hydrogel patch onto the heater. Turning up the temperature triggered a phase change in the patch, releasing the antibiotics.
More information: Paramita Das et al. Nacre Mimetic with Embedded Silver Nanowire for Resistive Heating, ACS Applied Nano Materials (2018). DOI: 10.1021/acsanm.7b00348
Abstract
Nacre mimetics show great potential as lightweight, mechanically robust, and functional materials. Here, we introduce highly reinforced nacre-mimetic nanocomposite (NC), prepared via self-assembly of poly(vinyl alcohol) polymer-coated synthetic nanoclay from aqueous dispersions, as a transparent and mechanically robust substrate to prepare silver nanowire (AgNW) embedded thin-film resistive heater. AgNW is a promising substitute for widely used brittle and expensive indium tin oxide (ITO) due to its high electrical conductivity, excellent optical transparency, and good mechanical flexibility. However, current AgNW-based electrodes mostly suffer from limitations such as surface roughness and weak adhesion of AgNWs to flexible plastic substrates (e.g., poly(ethylene terephthalate), PET). We demonstrate that AgNW can be embedded in nacre-mimetic NC substrate by simple hot-pressing as compared to PET, leading to excellent stability against scotch tape peeling without any encapsulation layer. The AgNW/NC resistive heater shows uniform sheet resistance varying from 10 to 80 Ω/sq with 70 to 91% transmittance at 550 nm on decreasing AgNW density from 111 to 23 mg/m2. Moreover, AgNW/NC heater can generate rapid (10 s) and repetitive long-term heating response at low input voltages. It shows only small variation in temperature (4 °C) and sheet resistance during bending at 14 mm diameter for 2000 cycles as well as under various extreme mechanical deformations. Taking advantage of the conformability, mechanical deformability, and fast thermal response of the resistive heater, we demonstrated the in vitro temperature-triggered release of antibiotics (i.e.,vancomycin) from phase change material-coated, antibiotic-loaded hydrogels. Hence, we envision that the resistive heater can be introduced as a potential candidate into flexible electronics and wearable devices.
Provided by American Chemical Society