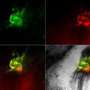
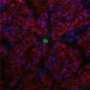
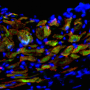
Live 3-D imaging highlights cellular activity during embryonic heart development
Live imaging techniques have given Spanish researchers deeper insight into the development of the embryonic heart in mice. Their analysis reveals the coordination that occurs between cardiac progenitor cells – cells similar to stem cells that can change into another specific type of cell during heart development. They found that progenitor cells go through alternate phases of changing into cardiac muscle (differentiation) to initiate early heart function, and contribute to cardiac morphogenesis. The study, which was originally published as a preprint in bioRxiv, appears today in the journal eLife.
"Previous studies have shown that two populations of progenitor cells are involved in vertebrate heart development: first heart field (FHF) cells and second heart field (SHF) cells," explains lead author Kenzo Ivanovitch of the Spanish National Cardiovascular Research Center (CNIC). "FHFs are recruited early in development to form the initial shape of the heart tube, and SHFs are brought in later to extend the length of the tube. However, until now, both the exact role of cell differentiation and cellular movements during heart development have not been captured."
To look further into this process, Ivanovitch and his team, including Susana Temiño, a technician at CNIC, established a whole-embryo, live-imaging method based on two-photon microscopy that allows tracking tissues at the cellular level. Using various genetic tracing tools, they labeled progenitor and differentiated cells and tracked them in 3-D over time. They then combined these images with 3-D reconstructions of the heart tube at multiple stages of development.
"Our images showed three distinct phases of heart tube formation," explains Ivanovitch. "During an initial phase, FHF cells differentiate rapidly to form a cardiac crescent, while limited morphogenesis takes place. In the second stage, there are no differentiation events, but extensive morphogenesis results in the fully formed heart tube. And in the third and final phase, cardiac cell differentiation resumes and contributes to the known SHF-derived regions of the heart. This tightly regulated schedule allows the young heart to start working as the embryo requires, while simultaneously building the complex architecture of the final, definitive organ."
These findings highlight the essential role that FHF and SHF cells play in embryonic heart development and reveal tissue-level coordination between alternating phases of differentiation and morphogenesis during the formation of the heart tube.
Senior author Miguel Torres, Group Leader at CNIC, says, "In the future, we hope that building on this work will help reveal new mechanisms of organogenesis. This would have important implications for greater understanding of both normal heart development and the origins of abnormalities in newborn babies."
More information: The paper 'Live imaging of heart tube development in mouse reveals alternating phases of cardiac differentiation and morphogenesis' can be freely accessed online at doi.org/10.7554/eLife.30668.
This study originally appeared as a preprint in bioRxiv, at doi.org/10.1101/170522.
Journal information: eLife
Provided by The Centro Nacional de Investigaciones Cardiovasculares