Designer biosensor can detect antibiotic production by microbes
Researchers from North Carolina State University have engineered designer biosensors that can detect antibiotic molecules of interest. The biosensors are a first step toward creating antibiotic-producing "factories" within microbes such as E. coli.
Macrolides are a group of naturally occurring small molecules that can have antibiotic, antifungal or anticancer effects. The antibiotic erythromycin is one example - it is a macrolide produced by soil-dwelling bacteria. Researchers are interested in using these natural antibiotics and the microbes that produce them in order to develop new antibiotics; however, microbes that produce antibiotic macrolides only make small amounts of a limited variety of antibiotics.
"Our ultimate goal is to engineer microbes to make new versions of these antibiotics for our use, which will drastically reduce the amount of time and money necessary for new drug testing and development," says Gavin Williams, associate professor of bio-organic chemistry at NC State and corresponding author of a paper describing the research. "In order to do that, we first need to be able to detect the antibiotic molecules of interest produced by the microbes."
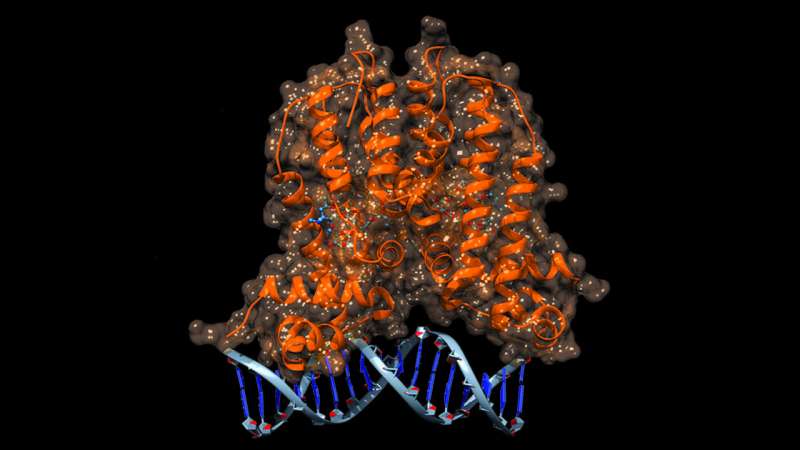
Williams and his team used a naturally occurring molecular switch - a protein called MphR - as their biosensor. In E. coli, MphR can detect the presence of macrolide antibiotics being secreted by microbes that are attacking E. coli. When MphR senses the antibiotic, it turns on a resistance mechanism to negate the antibiotic's effects.
The researchers created a large library of MphR protein variants and screened them for the ability to switch on production of a fluorescent green protein when they were in the presence of a desired macrolide. They tested the variants against erythromycin, which MphR already recognizes, and found that some of the MphR variants improved their detection ability tenfold. They also successfully tested the variants against macrolides that were not closely related to erythromycin, such as tylosin.
"Essentially we have co-opted and evolved the MphR sensor system, increasing its sensitivity in recognizing the molecules that we're interested in," says Williams. "We know that we can tailor this biosensor and that it will detect the molecules we're interested in, which will enable us to screen millions of different strains quickly. This is the first step toward high-throughput engineering of antibiotics, where we create vast libraries of genetically modified strains and variants of microbes in order to find the few strains and variants that produce the desired molecule in the desired yield."
The research appears in ACS Synthetic Biology, and was funded by the National Institutes of Health (grant GM104258) and the NC State Chancellor's Innovation Fund. Graduate student Yiwei Li, former graduate student Christian Kasey, former undergraduate student Mounir Zerrad, and T. Ashton Cropp, professor of chemistry at Virginia Commonwealth University, contributed to the work.
More information: Christian Kasey et al, Development of transcription factor-based designer macrolide biosensors for metabolic engineering and synthetic biology, ACS Synthetic Biology (2017). DOI: 10.1021/acssynbio.7b00287
Journal information: ACS Synthetic Biology
Provided by North Carolina State University