Gene editing used to eliminate viruses in live pigs
eGenesis, a biotechnology company focused on transforming xenotransplantation into a lifesaving medical procedure, announced the publication of a study in the journal Science by eGenesis scientists and their collaborators demonstrating the inactivation of PERV to prevent cross-species viral transmission and a breakthrough in producing the first PERV-free pigs, an important milestone for xenotransplantation.
Xenotransplantation, the use of animal organs for human transplant, is a promising approach to alleviate the severe shortage of organs for human transplantation but the risk of cross-species transmission of PERVs, among other issues, has to date impeded its use in humans. eGenesis is committed to harnessing CRISPR technology to deliver safe and effective human transplantable cells, tissues and organs grown in pigs, thus addressing a dire need for hundreds of thousands of patients worldwide.
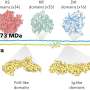
"This is the first publication to report on PERV-free pig production," said Luhan Yang, Ph.D., co-founder and chief scientific officer at eGenesis. "We generated a protocol to enable multiplex genome editing, eradicated all PERV activity using CRISPR technology in cloneable primary porcine fibroblasts and successfully produced PERV-free piglets. This research represents an important advance in addressing safety concerns about cross-species viral transmission. Our team will further engineer the PERV-free pig strain to deliver safe and effective xenotransplantation."
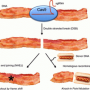
This study examined the risk of PERV infectivity and demonstrated in vitro that PERVs infected human cells and were transmitted to human cells that had no history of contact with porcine cells, substantiating the need to address this issue in order to ensure safe xenotransplantation practice.
Researchers developed a strategy to enable efficient and precise genome editing in primary cells using CRISPR-Cas9 technology. In conjunction with a method to inhibit primary cell death during multiplex genome editing, researchers successfully produced viable PERV-free porcine embryos via somatic cell nuclear transfer, using engineered primary cells. They then implanted the PERV-free embryos into surrogate sows and demonstrated the absence of PERV re-infection, initially in fetuses and finally in recently born piglets. These piglets are the first animals born free of endogenous virus and will be monitored for any long-term effects and impact by the eGenesis team.
The eGenesis team, having produced the first piglets free of active PERVs, is working toward combining the safety benefits of PERV-free pigs with additional gene editing addressing immunological response to increase organ immune and functional compatibilities.
More information: D. Niu el al., "Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9," Science (2017). science.sciencemag.org/lookup/ … 1126/science.aan4187
Journal information: Science
Provided by eGenesis