2-D materials clean up their act
Two-dimensional materials such as graphene may only be one or two atoms thick but they are poised to power flexible electronics, revolutionise composites and even clean our water.
However, being this thin comes at a price: the functional properties we depend on will change if the material becomes contaminated.
Luckily, many 2-D materials exhibit the 'self-cleaning phenomenon', meaning when different 2-D materials are pressed together, stray molecules from the air and the lab are pushed out leaving large areas clear of impurities.
Since graphene's isolation in 2004 a whole host of other 2-D materials have been discovered each with a range of different properties.
When graphene and other 2-D materials are combined, the potential of these new materials comes alive.
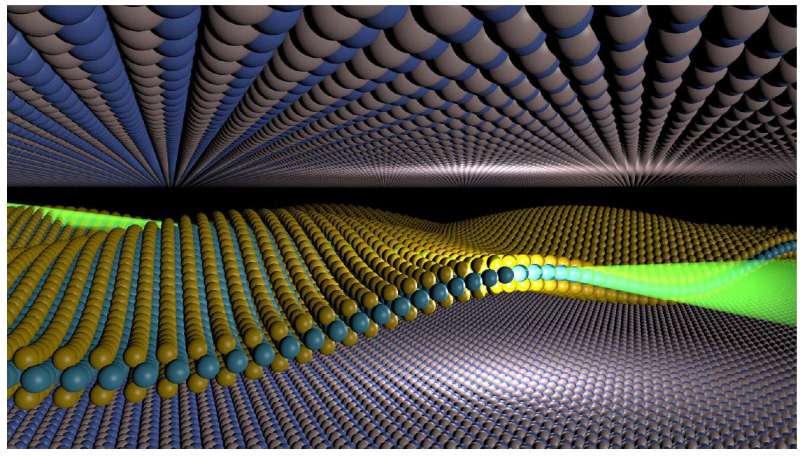
Layering stacks of 2-D materials in a precisely chosen sequence can produce new materials called heterostructures that can be fine-tuned to achieve a specific purpose (from LEDs, to water purification, to high speed electronics).
These flat regions have yielded some of the most fascinating physics of our time. Now, the assumption that these areas are completely clean is under scrutiny.
Writing in Nano Letters a team of researchers at the National Graphene Institute at The University of Manchester have shown that even the gas within which the 2-D material stacks are assembled can affect the structure and properties of the materials.
They found that for one class of 2-D materials called the transition metal dichalcogenides (TMDCs), some had a very large gap between them and their neighbour; a distance unexplained by theoretical calculations done by Professsor Katsnelson and Dr Rudenko at Radboud University, Netherlands.
These observations all seemed to point to the presence of impurities between the 2-D materials. To confirm this, 2-D materials were stacked in a pure Argon gas atmosphere using a sealed chamber (known as a glove-box) in which the environment can be completely controlled.
Where previously the same material had given large gaps between neighbours, this time gave distances matching those predicted by theory for a clean interface free from impurities.
Dr Aidan Rooney, who imaged the structures using high resolution electron microscopy, explained:
"By taking a side view of these sandwich structures we can see how these unique materials stick together and discover new secrets we have previously missed."
Dr Sarah Haigh, who led the team of researchers who carried out this work said:
"This sort of insight is changing how we build devices like LEDs and sensors from 2-D materials. The properties of these devices were known to depend heavily on how and where we make them, and for the first time we have observed why."
The consequences of this finding will directly impact on how we make graphene devices for future applications, showing that even the environment within which 2-D material stacks are assembled affects the atomic structure and properties.
More information: Aidan. P. Rooney et al. Observing Imperfection in Atomic Interfaces for van der Waals Heterostructures, Nano Letters (2017). DOI: 10.1021/acs.nanolett.7b01248
Journal information: Nano Letters
Provided by University of Manchester