August 3, 2017 report
Chemotactic movement used to carry drugs through blood-brain barrier
(Phys.org)—An international team of researchers has developed a way to take advantage of chemotactic movement to carry drugs through the blood-brain barrier. In their paper published on the open access site Sciences Advances, the researchers describe the technique and its effectiveness in test rats.
Medical scientists have worked for many years to find medications to treat brain ailments, many of which have proven effective—but their use has been stymied by the blood brain-barrier. The blood-brain barrier is a filtering mechanism involved with capillaries that carry blood to the brain. The filter is adapted to prevent harmful chemicals from entering one of the most critical organs. Scientists have looked at a variety of ways to coax chemicals through the blood-brain barrier, and while some have met with success, there is still a great need for improved options. In this new effort, the researchers have found a way to use chemotactic movement to carry desired chemicals through the filter.
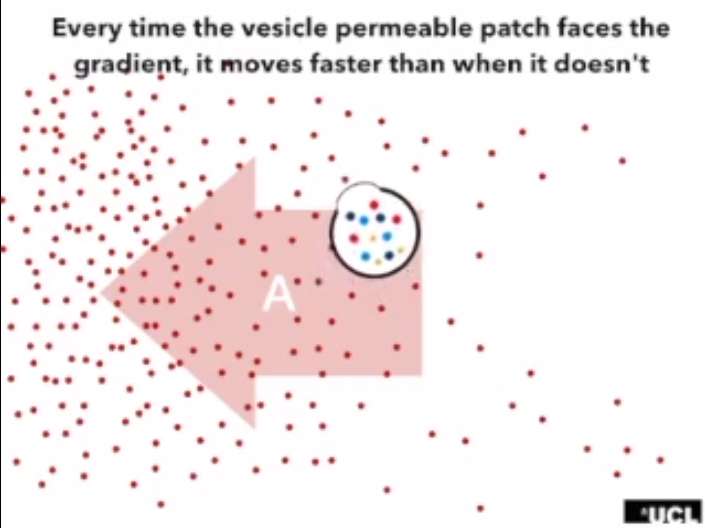
Chemotactic movement occurs when an organism responds to chemicals in the environment, causing them to move. The new technique does not involve inserting a foreign organism into the blood stream, of course; instead, it involves injecting vesicles (dubbed "nanoswimmers"), which are little pods filled with chemicals that move on their own through a chemical action called Brownian motion. Such motion is induced by making the vesicles asymmetric, with one side more permeable than the rest of the vesicle shell. This allows more of the material inside to react with material outside at just one side of the vesicle, causing it to move toward the material it finds attractive.
The researchers noted that one of the main materials that passes through the blood brain barrier is glucose, because the brain uses it as an energy source. By causing the vesicles to be attracted to glucose in higher concentrations, the researchers were able to coax the vesicle through the blood-brain barrier and into the brains of test mice. Once there, the vesicles were free to dissolve, releasing the chemicals inside. The researchers report finding a four-fold increase in the amount of medicine reaching the brain as compared to conventional methods.
More information: Adrian Joseph et al. Chemotactic synthetic vesicles: Design and applications in blood-brain barrier crossing, Science Advances (2017). DOI: 10.1126/sciadv.1700362
Abstract
In recent years, scientists have created artificial microscopic and nanoscopic self-propelling particles, often referred to as nano- or microswimmers, capable of mimicking biological locomotion and taxis. This active diffusion enables the engineering of complex operations that so far have not been possible at the micro- and nanoscale. One of the most promising tasks is the ability to engineer nanocarriers that can autonomously navigate within tissues and organs, accessing nearly every site of the human body guided by endogenous chemical gradients. We report a fully synthetic, organic, nanoscopic system that exhibits attractive chemotaxis driven by enzymatic conversion of glucose. We achieve this by encapsulating glucose oxidase alone or in combination with catalase into nanoscopic and biocompatible asymmetric polymer vesicles (known as polymersomes). We show that these vesicles self-propel in response to an external gradient of glucose by inducing a slip velocity on their surface, which makes them move in an extremely sensitive way toward higher-concentration regions. We finally demonstrate that the chemotactic behavior of these nanoswimmers, in combination with LRP-1 (low-density lipoprotein receptor–related protein 1) targeting, enables a fourfold increase in penetration to the brain compared to nonchemotactic systems.
Journal information: Science Advances
© 2017 Phys.org