Teaming nature and technology for climate solutions

One corner of the Omani desert is blanketed by a type of rock with an unquenchable thirst for a colorless and odorless gas vital to life on Earth. That gas is CO2, and when it reacts with peridotite, a rock abundant in the Earth's mantle, it's soaked up, forming a solid carbonate similar to limestone.
The Omani peridotite currently absorbs an estimated 10,000 to 100,000 tons of carbon dioxide per year, but scientists say that with a little human intervention, it could be sped up to absorb one-eighth of the 38 billion tons of CO2 emitted through the burning of fossil fuels around the world. A greenhouse gas, CO2 accumulates in the Earth's atmosphere, where it traps heat and raises the global average temperature, fueling extreme weather such as hotter heat waves, more frequent droughts, and more powerful hurricanes. The current concentration of CO2 is around 400 ppm, the highest it's been in at least the last 800,000 years.
Although it's just one of the rock types with CO2-absorbing properties, and just one method of reducing the impact of CO2 emissions, peridotite could help mitigate the impending risks posed by climate change.
The pioneers behind the peridotite research, Peter Kelemen and Juerg Matter, geologists at Columbia University's Lamont-Doherty Earth Observatory, discovered peridotite's affinity for consuming CO2 when they brought it into the lab to determine its age. Realizing that the peridotite had reacted with CO2 relatively recently, they began to conceptualize what scaling the reaction might look like.
Although it would be too expensive to move the rock close to power stations where it could soak up emissions, the researchers suggest that CO2 could be funneled into the Earth's peridotite-laden mantle through a process similar to hydraulic fracturing. This could open up a giant repository for the gas that wouldn't depend on location, but carries with it environmental implications that would need to be taken into careful consideration.
Nature-Driven Solutions
Geologists have long understood that rocks are an important carbon sink. Rock weathering happens when CO2 dissolves in droplets of rainwater, adding the acidity necessary to dissolve the minerals that make up the rock. Rock weathering pulls an estimated one billion tons of CO2 out of the atmosphere annually.
"Understanding these natural chemical processes can lead to breakthroughs that allow us to use and expedite processes that reduce CO2 in the atmosphere," says Bradley Sageman, professor and chair of earth and planetary sciences at Northwestern. "Methods like these that are standard tech today were considered science fiction in the past. Take the Omani peridotite example. If we could harness that reaction, we've got a potentially transformative mechanism to absorb CO2 at scale."
Some of Sageman's colleagues study the kinetics of weathering reactions to gain a fundamental understanding of the carbon cycle—the circular transformation of carbon between living things and the environment. Natural users of CO2 include forests, wetlands, and peat bogs. Scientists have studied these and other carbon sinks to develop many artificial processes that produce similar effects.
Two commonly discussed types of artificial sequestration are ocean storage—pumping CO2 deep into the ocean—and geologic sequestration—injecting CO2 deep into depleted oil and gas reservoirs or coal beds that can't be mined. Scientists hesitate to pursue either of these solutions aggressively out of concerns about the stability of disturbed natural systems and the potential effects on ocean life.
To better understand the dynamics of storage solutions, Sageman and his team look to periods in Earth's history characterized by high levels of atmospheric CO2 and warming. "A lot of the work we do is to improve our understanding of how the Earth's system behaved during past events of global warming. This should lead to a better framework for discerning what may happen in a future warming world," he says.
While the capture and long-term storage of CO2 suggest some viable solutions to mitigating CO2, scientists are also looking at the gas as a resource for creating clean energy. All around the world, researchers are demonstrating that CO2 can be a key ingredient in many technologies that produce clean, carbon-neutral energy.
Such technologies could complement current fossil fuel-based systems to lower emissions, and eventually capture CO2 from the atmosphere to help mitigate climate change. Companies big and small—across industries that range from energy to airlines to automotive—are taking notice.
Industrial Strength Solutions
Industry strategists across the board, from small startups to multi-national companies, are looking to define their roles and opportunities in a clean energy future. They're looking for complementary skills, technologies, or technologists who are in the process of developing innovations that are technically feasible, but lack a view towards the market. They know that with risk comes reward, and the pioneers aren't waiting around for the perfect solution to their energy needs; they're working with the brightest technologists to create it.
Compared to companies in many other industries, utilities are not known to invest heavily in research and development, instead relying on a relatively static knowledge base. Disrupting that status quo, Exelon, the largest regulated utility in the country serving 10 million consumers, is aggressively investing in technologies it can shape into customer-facing products.
Exelon is investing in many early- and middle-stage projects that complement its services while also reducing its carbon footprint, including investments in solar energy, fuel cells, and batteries. One example, a company called NetPower, uses CO2 as a working fluid to drive a combustion turbine that generates electricity without producing any emissions. The system also produces pipeline-quality CO2 that can be stored or used in industrial processes, including an enhanced oil recovery process where CO2 is injected into an oil reservoir to boost output.
In March 2016, NetPower broke ground on a 50-megawatt demonstration plant in La Porte, Texas, with the goal of operating as efficiently as the best natural gas plants today. Part of a $140 million program, the plant will include ongoing technology advancement, a full testing and operations program, and commercial product development. Toshiba will provide a supercritical CO2 turbine and combustor for the project.
"A lot of people say natural gas is a bridge fuel to lower emissions in the electricity sector, but because most natural gas plants are powered by turbines that rely on a traditional steam cycle, they cannot produce high quality CO2 that can be repurposed for other things," says Gould. "On top of that, since NetPower plants don't require steam to drive their turbines, it eliminates water usage as well."
Fueling Value Creation
Like NetPower, many technology companies have developed processes to help industries reduce their carbon footprint, and in some cases, make new products in the process. One such company, LanzaTech, is making waves in carbon recycling with a proprietary biological process that uses a microbe to convert industrial emissions to useful fuels and chemicals.

"We convert emissions into a variety of new valuable products that would otherwise come from commodity feedstocks," says Prabhakar Nair, LanzaTech's vice president for business development.
LanzaTech's process works with a variety of microbes, allowing a customer to specify the desired output—currently either ethanol or butanediol—and take advantage of market conditions.
After opening two pre-production plants in China, LanzaTech plans to open its first scaled commercial plant in Shanghai in late 2017. The company is also working with the world's largest steel producer, ArcelorMittal, to implement a commercial-scale project at its flagship steel mill in Belgium.
The key to the company's success, according to Nair, lies in the synergy between the technology, industry partners, and product off-takers. Full service at a demo-scale facility includes linking industry partners with buyers for the byproduct that's produced there. For example, LanzaTech has linked steel producers to local refineries that are required by regulation to mix ethanol into their fuel blends.
"By serving as the bridge between industries that have a feedstock supply and those that have a need, and by doing that with waste emissions, we are putting the circular economy in motion," says Nair.
The company recently received $4 million from the US Department of Energy's Bioenergy Technologies Office to design and plan a demonstration-scale facility using industrial off-gases from steel manufacturing to produce three million gallons of low-carbon jet and diesel fuels a year. This comes on the heels of a partnership with Virgin Atlantic, which plans a test flight in 2017 using jet fuel made from LanzaTech's proprietary low-carbon ethanol. LanzaTech estimates that its technology is compatible with 65 percent of steel mills, and if implemented could produce 15 billion gallons of jet fuel a year, or one-fifth of aviation fuel used around the world.
The Race to the Solar Refinery
Imagine pulling up to the gas station tomorrow, but instead of choosing between unleaded, plus, or diesel, you reach for a highly efficient fuel made only from sunlight, water, and CO2.
These very components that make up this "solar fuel" are the same three things that living plants convert into food. Dubbed "artificial photosynthesis," at scale this process could jump major hurdles in reducing dependency on fossil fuels.
With its advantages, it's no wonder that solar fuels research has taken off around the world—from hubs in Japan and Sweden to the Joint Center for Artificial Photosynthesis (JCAP) in California, founded by the US Department of Energy in 2010. With a budget of $122 million over five years, JCAP's mission is to build a prototype of a solar fuels system.
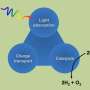
The components of the system envisioned at JCAP are fairly basic. The system requires a photovoltaic material to absorb light energy from the sun, which is then directed at two separate catalysts to lower the energetic hurdle for a reaction. One catalyst splits water into protons and oxygen and the other converts carbon dioxide and protons into hydrocarbons, the main components of fuels. While these processes are currently possible, they still present challenges.
One is economic. The materials used in both the photovoltaics and catalysts are expensive, including rare materials such as iridium or platinum, which presents a scalability challenge. The other is efficiency. Although ten times more efficient than natural photosynthesis at capturing and converting the sun's energy, the highest recorded efficiency for artificial photosynthesis is still only 10 percent. That's less than half the efficiency of silicon panels on the market today.
So why all the fuss to make energy-dense fuels when we have more efficient renewable technologies available today? Sources of renewable energy, including solar and wind, can only be generated intermittently—when the sun shines or wind blows. Fuels present a viable option for grid-scale energy storage that could compensate for that intermittency and be easily transported to where they're needed.
The energy density of fuels is also around 100 times greater than that of the highest performing batteries, and many methods of transportation—including cars, ships, and airplanes—already have the infrastructure to run on fuels. And when it comes to mitigating the effects of climate change, if these fuels were made from carbon dioxide captured from the air, the process would be carbon-neutral and would not emit new greenhouse gases into the atmosphere.
Collaboration on a Global Scale
Because many questions remain unanswered, JCAP has modified its goal of creating a solar fuels system, instead focusing on getting the fundamentals right. Meanwhile, other scientists carry on a whole system approach.
That approach will take collaboration and systems engineering, says Michael R. Wasielewski, Clare Hamilton Hall Professor of Chemistry and director of the Argonne-Northwestern Solar Energy Research (ANSER) Center. "Researchers have subsystems that can perform on some baseline level, but when you try to integrate them, it's not seamless and therefore not commercially viable. You need scientists collaborating with engineers to work out the bugs and create a complete working system," Wasielewski says.
At ANSER, an Energy Frontier Research Center of the US Department of Energy, Wasielewski works with more than 60 researchers to develop a fundamental understanding of the molecules, materials, and methods required to create significantly more efficient technologies for solar fuels and solar electricity production. In 2013, Wasielewski also founded the Solar Fuels Institute (SOFI).
SOFI launched a six-phase demonstration project in 2016 with sights set on taking a systems approach to developing solar fuels. At the end of last year, SOFI scientists had successfully produced methanol in the lab at Northwestern. "The SOFI demo project was viewed as a system from the outset," says Wasielewski. "We're going to need to make this work from one end to the other. But we can't do it alone."
As a global consortium, SOFI has university and industry partners from around the world—from academic institutions that span three continents to major multinationals like Shell and Total. Still, SOFI is seeking collaborations from wide-ranging fields, including economics and policy, to work on implementation strategies.
"Generally speaking," Wasielewski says, "scientists and engineers can come up with more than one solution to a problem. We can fine-tune a process to be that much more efficient, but we can't know what the priorities are for real-world customers if we don't involve them early on. That's what constitutes a breakthrough technology."
Provided by Northwestern University