Dynamic DNA helps ward off gene damage, study reveals
Researchers have identified properties in DNA's protective structure that could transform the way scientists think about the human genome.
Molecules involved in DNA's supportive scaffolding—once thought to be fixed—go through dynamic and responsive changes to shield against mutations, the research shows.
Experts say this finding is crucial to understanding DNA damage and genome organisation and could impact current thinking on DNA-linked diseases, including cancers.
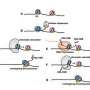
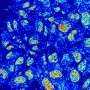
In human cells, DNA is wrapped around proteins to form chromatin. Chromatin shields DNA from damage and regulates what genetic information can be read—a process known as transcription.
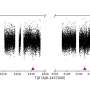
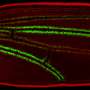
Researchers—led by the University of Edinburgh—showed that a chemical called scaffold attachment factor A (SAF-A) binds to specific molecules known as caRNAs to form a protective chromatin mesh.
For the first time, this mesh was shown to be dynamic, assembling and disassembling and allowing the structure to be flexible and responsive to cell signals.
In addition, loss of SAF-A was found to lead to abnormal folding of DNA and to promote damage to the genome.
SAF-A has previously been shown in mouse studies to be essential to embryo development and mutations of the SAF-A gene have repeatedly been found in cancer gene screening studies.
Scientists say the findings shed light on how chromatin protects DNA from high numbers of harmful mutations, a condition known as genetic instability.
The study—published in Cell—was carried out in collaboration with Heriot Watt University. It was funded by the Medical Research Council (MRC).
Nick Gilbert, Professor of Genetics at the University of Edinburgh's MRC Institute of Genetics and Molecular Medicine, said: "These findings are very exciting and have fundamental implications for how we understand our own DNA, showing that chromatin is the true guardian of the genome. The results open new possibilities for investigating how we might protect against DNA mutations that we see in diseases like cancer."
Cutting-edge techniques used in the study were developed by the Edinburgh Super-Resolution Imaging Consortium, which is supported by the MRC, the Biotechnology and Biological Sciences Research Council and the Engineering and Physical Sciences Research Council.
Professor Rory Duncan, Head of the Institute for Biological Chemistry, Biophysics and Bioengineering at Heriot-Watt University said: "The molecules involved in this study are as small to humans as Jupiter is large. The bespoke microscope techniques that we developed to understand these very tiny structures are important not only for this project but for all of biology."
Journal information: Cell
Provided by University of Edinburgh