May 26, 2017 report
The high cost of communication among social bees

(Phys.org)—Eusocial insects are predominantly dependent on chemosensory communication to coordinate social organization and define group membership. As the social complexity of a species increases, individual members require a greater diversity of signals. The communications of highly social insects such as wasps are well documented, but relatively little is known about the evolutionary transition between solitary and social living.
An international collaborative of researchers recently conducted a study of halictid bees in order to determine how the metabolic cost of chemosensory organs changes during such transitions. They theorized that changes in social structure would be reflected in changes to the expensive antennae sensory systems of insects.
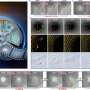
The researchers imaged the antennae of adult females from 36 species that ranged across a spectrum from completely solitary to highly social, and analyzed sensilla density using digital statistical software. The tests confirmed their theory: As sociality is gained and lost in halictid bees, convergent changes occur in both sensilla structures and the chemical signals of the groups. Social insects invest more in these systems than solitary bees, and as group complexity increases, communications develop higher diversity. "Taken together," the authors write, "these results suggest that there is a strong link between the evolution of social behavior and investment in communication."
The researchers note that sensilla density did not increase as sociality was gained; ancestrally solitary halictids had sensilla densities similar to eusocial species. The researchers compare this seeming paradox to the evolution of cave-dwelling animal species that lose the sense of vision, but whose visual organs and brain structures, while functionally redundant, never become completely vestigial.
However, secondarily solitary halictids—those bees that switched rapidly from social to solitary—exhibited marked decreases in sensilla density as communications declined. "Importantly, the reduction we observed in this group is not a complete vestigilization. Instead, it represents a decreased investment in antennal sensilla, perhaps in the absence of complex communication associated with group living," the authors write.
The authors suggest that higher sensilla density on the antennae of social insects confers two advantages: The increased surface area increases the speed of discrimination, allowing bees to quickly identify non-nestmates and more quickly respond with aggression. And the higher density may also increase the variety of chemoreceptors on the antennae, accommodating more precise signaling between nestmates. They also note that the higher densities of sensilla may include mechanosensory receptors, though they were unable to distinguish between the two types. It is true, however, that physical communication occurs among eusocial bee species, including bumping and ramming behaviors used by queens to induce submission in workers.
The researchers observe that investing in higher numbers of sensilla confers speed and accuracy in communication among social bees, and that while solitary females still recognize relevant stimuli, they no longer require metabolically costly sensilla for survival. "In conclusion," the authors write, "the rapid changes in chemical signaling within a single, polymorphous species, coupled with the repeated reduction in features associated with signal perception within and between species, suggest that there are trade-offs associated with communication in social groups."
Press release from Princeton University.
More information: Solitary bees reduce investment in communication compared with their social relatives. PNAS 2017 ; published ahead of print May 22, 2017, DOI: 10.1073/pnas.1620780114
Abstract
Social animals must communicate to define group membership and coordinate social organization. For social insects, communication is predominantly mediated through chemical signals, and as social complexity increases, so does the requirement for a greater diversity of signals. This relationship is particularly true for advanced eusocial insects, including ants, bees, and wasps, whose chemical communication systems have been well-characterized. However, we know surprisingly little about how these communication systems evolve during the transition between solitary and group living. Here, we demonstrate that the sensory systems associated with signal perception are evolutionarily labile. In particular, we show that differences in signal production and perception are tightly associated with changes in social behavior in halictid bees. Our results suggest that social species require a greater investment in communication than their solitary counterparts and that species that have reverted from eusociality to solitary living have repeatedly reduced investment in these potentially costly sensory perception systems.
Journal information: Proceedings of the National Academy of Sciences
© 2017 Phys.org