Clean technology to make low molecular weight chitosan

Researchers at MIPT have developed a new technique for obtaining low-molecular-weight, water-soluble chitin and chitosan. The proposed method relies on chitin and chitosan degradation by electron-beam plasma in a special plasma chemical reactor. The new technology reduces the time needed to produce water-soluble oligosaccharides of chitin and chitosan from several days to minutes. It also has the advantage of being environmentally friendly. The proposed method yields biologically active oligosaccharides with anti-microbial and fungicidal properties. The research findings were published in Carbohydrate Polymers.
Chitin is the second most abundant biopolymer after cellulose. Both chitin and its derivative chitosan were discovered about 200 years ago. However, in the last two decades, the two compounds have received increasing attention. In the natural world, chitin and chitosan occur as the main components of the exoskeletons of insects and crustaceans, as well as most fungi and some algae.
There are currently over 70 known applications of these compounds across many industries including agriculture, medicine, food processing, and manufacturing of cosmetics. Water-soluble chitooligosaccharides with low molecular weight rank among the most promising chitin- and chitosan-based products. They are conventionally produced by chemical depolymerization of the source material. This technology involves high temperatures and the use of hydrogen peroxide, concentrated solutions of organic and inorganic acids and sodium hydroxide, as well as other aggressive agents. Chemical treatment of chitin and chitosan results in large volumes of industrial wastewater with acid or alkali content that requires purification. Aside from toxic waste management, the conventionally employed chemical hydrolysis is time-consuming, a multistage process that may take up to several days.
MIPT's researchers teamed up with their colleagues from Lomonosov Northern (Arctic) Federal University to develop an entirely new and clean plasma-assisted method of producing low-molecular-weight derivatives of chitin and chitosan.
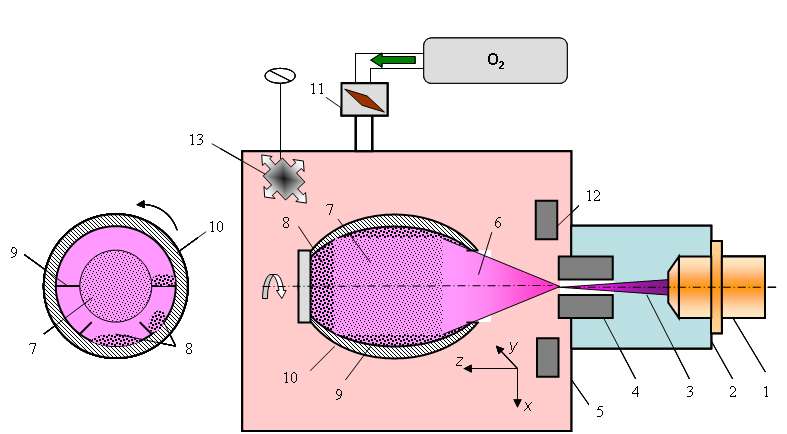
Electron-beam plasmachemical reactor
The scientists proposed an alternative plasma chemical technology to be used in place of chemical hydrolysis of chitin and chitosan. This promising technique involves the use of a low-temperature, nonequilibrium electron-beam plasma (EBP). To test the new technique, they introduced polysaccharide powder into their custom-built plasma chemical electron-beam reactor. While many gases could be used to fill the reaction chamber, oxygen and water vapor were found to be the most effective plasma-generating media for chitooligosaccharide production.
To generate plasma for chitin and chitosan treatment, a pre-relativistic electron beam was injected into the gaseous medium. Though the chamber contains gas, a high vacuum is required for electron beam generation. So the electron source must be screened by an injection window. When the electron beam passes through the medium, it causes ionization, excitation, and dissociation of gas molecules. As a result, radicals and other chemically active particles are obtained in very high concentrations that normally cannot be achieved under equilibrium conditions. Exposing chitin and chitosan to plasma and to the electron beam itself triggers the necessary biopolymer transformations. This happens without ever heating the polysaccharide powder above room temperature, preventing thermal destruction of the material. High temperatures are one of the main drawbacks of chemical hydrolysis.
Notably, the proposed technical solutions enable control over how much energy is released in the reaction medium, making the process stable and the plasma-treatment results replicable.
Biological activity of the compounds obtained
Practical applications of chitosan are determined by the compound's unique properties, namely its high biocompatibility, biodegradability, and complexation capacity combined with low toxicity. A number of studies carried out on E. coli, S. aureus, P. aeruginosa, S. enterica, B. subtilis, and some other species proved that the biological activity of chitosan depends significantly on its molecular weight. In particular, lower molecular weight chitosans were shown to inhibit bacterial growth and multiplication to a greater extent.
To evaluate the biological activity of the obtained oligosaccharides of chitin and chitosan, the researchers measured their anti-bacterial properties in vitro. The compounds were found to completely suppress the growth of both S. aureus and E. coli in replicating and resting forms. They also inhibited the growth of several species of filamentous fungi, namely P. tardum, P. chrysogenum, A. flavus, P. betae, and C. herbarum.
Tatiana Vasilieva, Ph.D. explains the significance of the study: "Our experiments have demonstrated that electron-beam plasma can be used in effective controlled chitin and chitosan depolymerization. This is an alternative method for obtaining low-molecular-weight, water-soluble, biologically active chitooligosaccharides. The proposed depolymerization technique can compete with the technologies conventionally employed by the chemical and biotech industries. Hopefully, these compounds will find their applications in agriculture, pharmaceutics, and medicine."
More information: T. Vasilieva et al, Formation of low molecular weight oligomers from chitin and chitosan stimulated by plasma-assisted processes, Carbohydrate Polymers (2017). DOI: 10.1016/j.carbpol.2017.01.026
Provided by Moscow Institute of Physics and Technology