Discovery of a facile process for H2 production using ammonia as a carrier
Hydrogen (H2) has attracted considerable attention as a clean energy source because the only by-product of its reaction with oxygen is water, and high efficiency for energy conversion is achieved when it is combined with fuel cell technologies.
However, low volumetric energy density and the dangers of transporting and handling H2 are drawbacks for commercial applications. These problems could be eliminated by using ammonia as a H2 storage medium (H2 carrier).
H2 produced from ammonia is utilized in fuel cells, engines, and turbines. However, the adoption of ammonia as a H2 carrier, especially for household and transportable devices, has been limited due to the lack of an efficient process for producing H2 and nitrogen by ammonia decomposition.
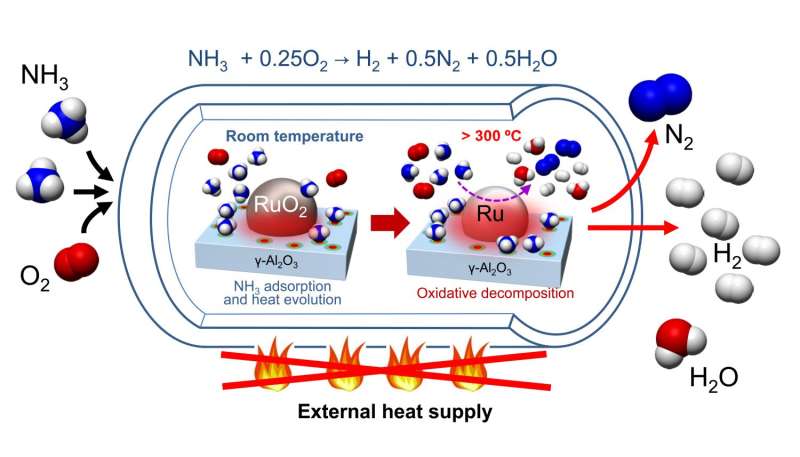
To overcome this limitation, the research team, led by Dr. Katsutoshi Nagaoka and Dr. Katsutoshi Sato, set out to develop a process that could be initiated rapidly, and that could produce H2 at a high rate without the need for external heat.
They found that H2 can be produced by supplying ammonia and oxygen at room temperature to a pre-treated RuO2/γ-Al2O3 catalyst without external heating. The heat evolves by ammonia adsorption onto this catalyst, increasing it to the catalytic auto-ignition temperature of ammonia. Subsequently, production of H2 by oxidative decomposition of ammonia begins. In this process, once the reaction is initiated, it can start again repeatedly even if there is no external heat supply because adsorbed ammonia is desorbed during the reaction.
Dr. Nagaoka said, "Our discovery utilizes a simple fundamental physicochemical process, namely adsorption, to operate a reaction with minimal energy input. We expect this to contribute to the development of efficient, carbon-free energy production and thus to global solutions for energy and climate crises."
Provided by Oita University