Unique genome architectures after fertilisation in single-cell embryos
Using a newly developed method, researchers at the Institute of Molecular Biotechnology of the Austrian Academy of Sciences (IMBA) have been able to shed light on the complexity of genome reorganization occurring during the first hours after fertilization in the single-cell mammalian embryo. Their findings have recently been published in the journal Nature. The team of researchers (from three continents) have discovered that the egg and sperm genomes that co-exist in the single-cell embryo or zygote have a unique structure compared to other interphase cells. Understanding this specialized chromatin "ground state" has the potential to provide insights into the yet mysterious process of epigenetic reprogramming to totipotency, the ability to give rise to all cell types.
Fusion of the egg and sperm, two highly differentiated cell types, leads to formation of the single-cell embryo or zygote. During the first hours after fertilization, the two separate genomes undergo reprogramming events that presumably function to erase the memory of the differentiated cell type and establish a state of totipotency. The mechanisms underlying totipotency remain poorly understood but are essential for generating a new organism from a fertilized egg.
A major advance in single-cell genomics
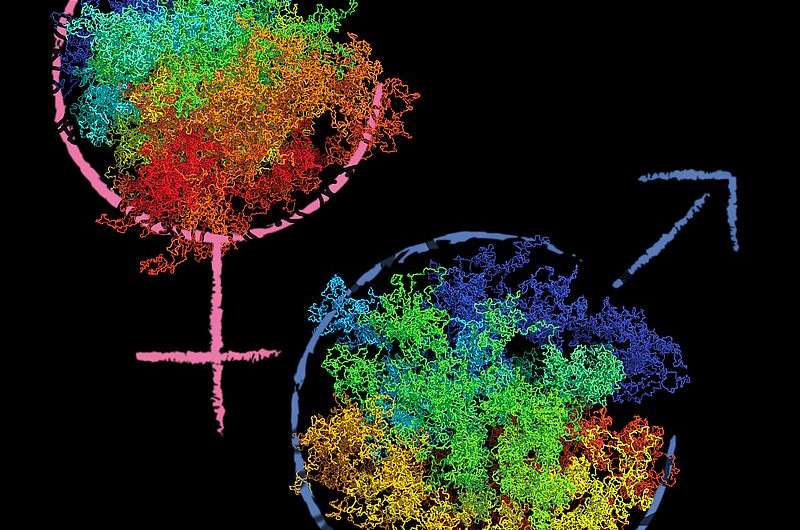
After fertilization, maternal and paternal genomes erase some of the epigenetic memory of the previously differentiated states in order to facilitate the beginning of new life as the zygote. In the first cell cycle after fertilization the maternal genome inherited from the oocyte (egg) and the paternal genome provided by sperm exist as separate nuclei in the zygote. The two genomes are marked by distinct epigenetic modifications acquired during reprogramming. Whether the 3-D chromatin structure of the maternal and paternal genomes is also distinct was not known.
An international team headed by Kikuë Tachibana-Konwalski from IMBA in collaboration with researchers from the Massachusetts Institute of Technology (MIT) in Boston and the Lomonosov Moscow State University (MSU) aimed to uncover how chromatin structure is reorganized during the mammalian oocyte-to-zygote transition. Using next-generation sequencing, bioinformatics analysis and mathematical modeling performed by Maxim Imakaev in Leonid Mirny's lab, the researchers identified specific patterns that emerge during genome reorganization in mouse oocytes and zygotes.
The low availability of starting material made it necessary to develop a new single-nucleus Hi-C (snHi-C) method that made it possible to analyze the chromatin architecture in oocytes and single-cell embryos for the first time. Using this method, features of genomic organization including compartments, topologically associating domains (TADs) and chromatin loops were detected in single cells when averaged over the genome. "Our method allowed us to detect chromatin contacts ten times more efficiently than a previous method. Because of this we were able to find differences in genome folding on the level of single cells: these cell-to-cell variations were missed in conventional Hi-C due to the averaging over millions of cells," says Ilya Flyamer, former Vienna Biocenter (VBC) summer student and then Master student and one of the first authors of the study.
Contrasting behaviour of maternal and paternal chromatin
"Additionally, we found unique differences in the three-dimensional organization of the zygote's chromatin compared to other interphase cells. What was even more interesting is that maternal and paternal genomes of the zygote seem to have different organizations within the same cell. It seems like the chromatin architecture is reorganized after fertilization, and that this reorganization happens differentially for the maternal and the paternal genomes," explained Johanna Gassler, PhD student at IMBA and one of the first authors of the study.
Senior author and IMBA group leader Kikuë Tachibana-Konwalski is fascinated by the secrets of the mammalian oocyte-to-zygote transition and has been studying the miracle of life, and in particular the very first molecular steps, for many years. She also hopes the findings will generate new insights for the emerging field of totipotency. "To place the power of the zygote into context: Reprogramming to pluripotency by the Yamanaka factors takes several days with limited efficiency, whilst reprogramming to totipotency occurs in the zygote within hours. How this is achieved remains one of the key unknowns in biology. By studying the chromatin state of zygotes, we aim to gain insights into this mechanism, which could also have applications for regenerative medicine," says Tachibana-Konwalski, underlining her excitement for the potential applications for her favourite research topic.
More information: Ilya M. Flyamer et al. Single-nucleus Hi-C reveals unique chromatin reorganization at oocyte-to-zygote transition, Nature (2017). DOI: 10.1038/nature21711
Journal information: Nature
Provided by Institute of Molecular Biotechnology