Toward a safer permanent hair dye that mimics melanin
Coloring hair has become a common practice, particularly for people who want to hide their graying locks. But an ingredient in many of today's commercial hair dyes has been linked to allergic reactions and skin irritation. Now scientists have developed a potentially safer alternative by mimicking the hair's natural color molecule: melanin. Their report appears in the journal ACS Biomaterials Science & Engineering.
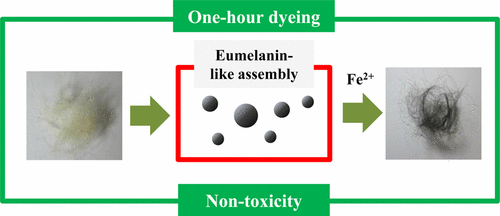
The permanent hair dye ingredient p-phenylenediamine (PPD) has been associated, although rarely, with allergic reactions including facial swelling and rashes. Coloring hair with natural melanin would be an intuitive alternative to PPD. But previous research has found that the pigment molecules clump together, forming rods and spheres too large to penetrate into the hair shaft to create lasting color. Jong-Rok Jeon and colleagues wanted to build on the idea of using melanin but with a molecule that mimics the real thing.
The researchers turned to polydopamine, a black substance that is structurally similar to melanin and has been explored for use in a variety of biomedical applications. Polydopamine with iron ions transformed gray hairs into black and lasted through three wash cycles. Lighter shades could also be achieved with polydopamine by pairing it with copper and aluminum ions. And toxicity tests showed that mice treated with the colorant didn't have noticeable side effects, while those that received a PPD-based dye developed bald spots.
More information: Kyung Min Im et al. Metal-Chelation-Assisted Deposition of Polydopamine on Human Hair: A Ready-to-Use Eumelanin-Based Hair Dyeing Methodology, ACS Biomaterials Science & Engineering (2017). DOI: 10.1021/acsbiomaterials.7b00031
Abstract
Permanent dyeing of gray hair has become an increasingly active area in the cosmetics industry because of the increasingly aging population in developed countries. So far, p-phenylenediamine (PPD) and related diamine-based monomeric compounds have been widely used for the dyeing processes, but toxicological studies have revealed such compounds to be carcinogenic and allergenic. Here, we for the first time demonstrated that polydopamine, a mimic of human eumelanin, gives rise within a commercially acceptable period of time (i.e., 1 h) to deep black colors (i.e., natural Asian hair colors) in human keratin hairs in the presence of ferrous ions. The dyed hairs showed excellent resistance to conventional detergents, and the detailed color was readily varied by changing the kind of metal ion used. SEM images and FT-IR-ATR spectra suggested that the extent of polydopamine aggregation was crucial for the dyeing efficiency. High-resolution (15 T) FT-ICR mass spectrometry performed on the products detached from hairs with either 0.1 N HCl or NaOH indicated that similar polydopamine products were recruited into the hair matrices whether in the presence or absence of metal-based chelating. Polydopamine chains were determined using EPR and ICP-OES to use chelation of ferrous ions to self-assemble as well as to bind keratin surfaces in the dyeing conditions. Also, mice subjected to skin toxicity tests showed much greater viability and much less hair loss with our dyeing agents than with PPD. In conclusion, this study showed that a safe eumelanin mimic may be used to permanently dye gray hair, and showed three kinds of deposition mechanisms (i.e., innate binding ability of polydopamine, metal-assisted self-assembly of polydopamine, and metal-related bridging between keratin surface and polydopamine) to be involved.
Provided by American Chemical Society

















