Primate-parasite network analyses show how germs jump from host to host

An extensive review of research on wild primate social networks and parasites underscores the importance of super-spreaders, or central individuals that play an outsized role in transmission of a pathogen.
Trends in Parasitology published the review, the first comprehensive synthesis of environmental and theoretical studies of disease dynamics in wild primate species—most of which are now threatened with extinction.
"This review gives critical insights that are applicable not just to conservation, but to our understanding of emerging infectious diseases and human health," says Thomas Gillespie, lead author of the review and a primate disease ecologist at Emory University. "We hope it helps jump-start a new way of approaching research into disease transmission - one that integrates ecology, behavior and evolution on a grand scale."
About 60 percent of the more than 500 known primate species face an extinction threat and more than three-fourths of them are on the decline, due to loss of habitat, hunting and disease. Large chunks of the world's forests are succumbing to agriculture, mining and logging, bringing people, primates, pets and livestock into closer proximity. That juxtaposition sets the stage for more pathogens to jump between humans and our closest relatives. More than 20 percent of wild primate species harbor parasites capable of spilling over into humans. HIV, Ebola, yellow fever and respiratory viruses are examples of viruses shared by humans and wild primates.
"How an emerging pathogen spreads through a species tends to be 'a black box' until it causes an outbreak among people," Gillespie says. The Zika virus, for instance, was first identified in monkeys in Uganda in 1947 but was not widely studied until recently, after it started sweeping through human populations.
Traditional infectious disease models generally assume that all individuals within a population have an equal probability of receiving or giving an infection, but more powerful analytical tools are showing that is not the case.
"Especially during the last decade," Gillespie says, "it's become much clearer that, just as everything in natural systems is heterogeneous, so is disease transmission."
The so-called 80-20 rule - 80 percent of disease transmission events in an epidemic are caused by 20 percent of individuals - is now a well-established phenomenon. What's not yet as well-understood are all of the variations within a disease system, and how these variations can combine to make an individual a super-spreader.
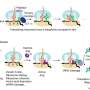
"Primate-parasite networks are an ideal model system for studying these questions," Gillespie says, "because primate species are so varied and they have sophisticated social structures. We also have behavioral data for many primates - detailing the hierarchical ranking of individuals, and whom those individuals are related to and whom they interact with - collected over decades of observational studies."
Combining knowledge of behaviors by individual species with data on their parasites can help researchers sort out high-risk traits and identify super-spreaders that contribute to transmission in a predictable way. Such patterns can help researchers to develop targeted interventions.
The review cites the example of a vaccine under development for Ebola virus, which is designed to replicate and spread immunity from a few individuals to others in a population. Targeting the most central individuals in an ape group with such a vaccine may be an effective way to mitigate future Ebola-related ape die-offs and spillover events into human populations.
The review also lays out a vision for the next decade of research into pathogen transmission, one that integrates ecology, behavior and evolution for studies of wild primates, other wildlife and humans.
"In addition to looking at how sociality influences infection, this review highlights the need to understand more about the flip side of that question: How do pathogens affect behavior and shape sociality?" Gillespie says. "We want to get people thinking about questions like this, in parallel networks of species."
More information: Julie Rushmore et al. Making New Connections: Insights from Primate–Parasite Networks, Trends in Parasitology (2017). DOI: 10.1016/j.pt.2017.01.013
Journal information: Trends in Parasitology
Provided by Emory University