Atomic map gives malaria drug new lease on life

Researchers have for the first time mapped how one of the longest-serving malaria drugs works, opening the possibility of altering its structure to make it more effective and combat increasing malaria drug resistance.
The study produced a precise atomic map of the frontline antimalarial drug mefloquine, showing how its structure could be tweaked to make it more effective in killing malaria parasites.
Dr Wilson Wong and Dr Brad Sleebs led the research at the Walter and Eliza Hall Institute, with Dr Jake Baum at Imperial College London, Dr Sjors Scheres from the Laboratory of Molecular Biology, Cambridge, and Dr Stuart Ralph at Melbourne's Bio21 Institute. The paper was published today in Nature Microbiology.
While drug treatments and preventative medicines for malaria are available, some have serious side effects and drug resistance means there is an urgent need for new treatments.
Many drugs that enter the market are known to kill harmful pathogens, but their exact mechanism for doing so is often unknown.
Dr Wong said it was the first time any team had identified one of the modes of action of mefloquine. "Mefloquine has been in use for more than 40 years, yet we did not understand how it was killing the parasite until now," he said.
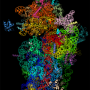
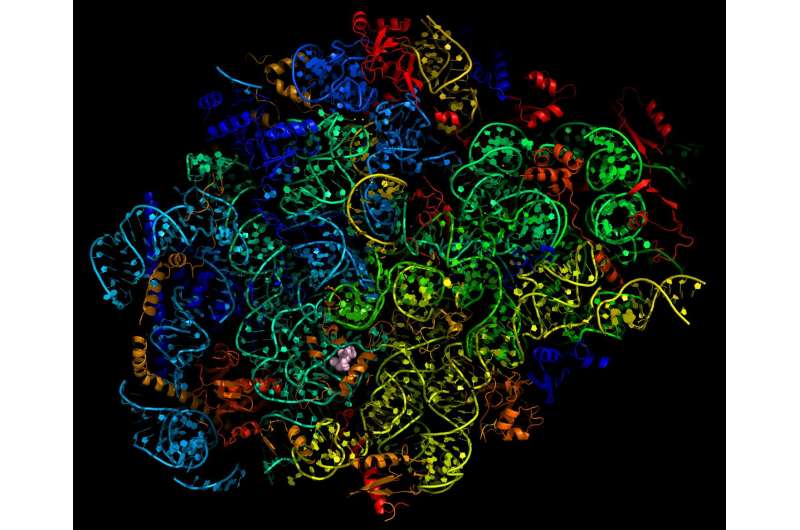
The team used cryo-electron microscopy, which produces images of biological molecules in their natural state in unprecedented detail, to see exactly how and where the drug binds the malaria parasite. "We discovered that mefloquine attacks the ribosome - the molecular machinery that manufactures proteins required for malaria parasite survival," Dr Wong said.
Mefloquine has been associated with some serious side effects, including neurological symptoms. While mefloquine's attack on the ribosome is analysed in this study, the authors point out that the drug likely targets other parts of the parasite too, killing it through multiple modes of action.
Dr Wong said the detailed atomic map would enable future drug improvements. "We now know mefloquine binds to a hotspot of activity on the ribosome surface," he said. "However our map of the ribosome and drug-binding site showed the fit is not perfect. We were able to mimic this interaction with compounds that were able to block the protein machinery and kill the parasite more effectively."
Dr Sleebs said using this information to redesign the drug to be more targeted and better differentiate between malaria and human ribosomes would be the next step. "If we could create a drug that targets this particular mode of action, it could be more efficient and potentially more safe for treating malaria."
Dr Jake Baum from Imperial College London said improving the action of existing drugs was a cheaper, quicker way of getting new treatments to patients. "It takes a lot of resources to discover new drugs, but by tweaking the structure of existing drugs we can breathe new life into them, and potentially gain significant benefits with far fewer resources," Dr Baum said. "With growing resistance to frontline antimalarial drugs, it makes sense to improve secondary drugs that work imperfectly, rather than always reinvent the wheel."
Cryo-electron microscopy continues to be a revolutionary technique to investigate the mode of action of new and existing therapies, and could be applied to many other drugs to help improve their design, or design completely new drugs de novo. Seen in this light, drug design with a microscope is likely to become a powerful tool for drug discovery for many of our most deadly pathogens.
More information: [52] Mefloquine targets the Plasmodium falciparum 80S ribosome to inhibit protein synthesis, Nature Microbiology, nature.com/articles/doi:10.1038/NMICROBIOL.2017.31
Journal information: Nature Microbiology
Provided by Walter and Eliza Hall Institute