Gold nano-antennas reveal single molecules' electrochemical properties
Individual molecules are extremely hard to see through feeble fluorescence. Leiden physicists have managed to use tiny gold nanorods as antennas to intensify their signal 500 times. They have published their results in Angewandte Chemie.
In the early 1980's, IBM created a world-famous picture of individual particles spelling out the company's name. They used a scanning tunneling microscope to image single particles for the first time. A decade later, scientists managed to see molecules using visible light by picking up on the molecules' fluorescence. Physicist Michel Orrit from Leiden University (Netherlands) was one of those pioneers.
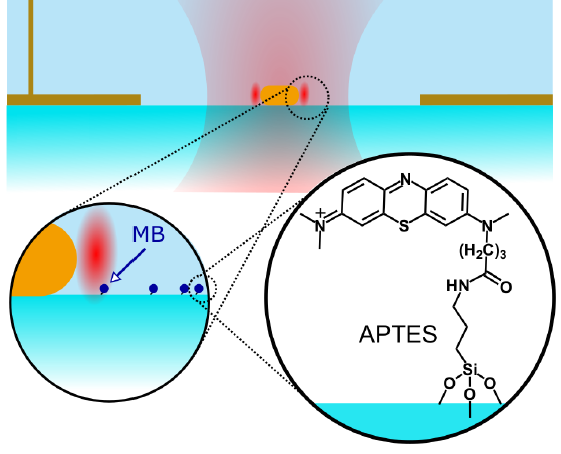
Now, Orrit and his group have further developed that research, using tiny antennas to amplify the signal for better visualization. Instead of a large metal antenna for radio waves, they used a small rod to catch visible light waves. This gold nanorod—40 x 80 nanometers in size—locally intensifies the electromagnetic field of light at both ends. If a molecule is situated at either end, it will fluoresce 500 times more strongly than without the rod.
Orrit's group published an article in Angewandte Chemie in which they described how they tested their concept on so-called redox reactions. These occur in any type of electric process, for example in solar cells. "We tested on redox reactions as a proof of concept," says Martin Caldarola, one of the authors. "At the same time, redox reactions are a very interesting application of our technique for many scientists, because they occur in so many processes."

Caldarola tested the technique by determining the redox midpoint potential—a crucial indicator for a material's ability to perform redox reactions. This offers, for example, its applicability for solar cells. The new method enables researchers to resolve the midpoint potential for each molecule, so they can investigate materials very precisely.
More information: M.Sc. Weichun Zhang, Dr. Martín Caldarola, M.Sc. Biswajit Pradhan, Prof. Dr. Michel Orrit, 'Gold Nanorod-Enhanced Fluorescence Enables Single-Molecule Electrochemistry of Methylene Blue', Angewandte Chemie
Journal information: Angewandte Chemie
Provided by Leiden Institute of Physics