February 3, 2017 report
Fluorescent probe seeks out pancreatic cancer cells in mice
(Phys.org)—A diagnosis of pancreatic cancer often means the patient has less than a year to live, and some reports suggest that pancreatic cancer will become the second-leading cause of cancer-related death in the next fifteen years. Patients often require surgery to remove the cancerous tissue as chemotherapy or radiation do not significantly improve patient prognosis. However, current methods to detect whether all of the cancerous tissue has been removed are either too slow to use during surgery or are inaccurate, risking incomplete removal of cancerous tissue.
In an effort to improve mid-surgery cancer tissue screening and pre-surgery detection, researchers from Portland State University and Oregon Health & Science University have developed a fluorescent probe that targets pancreatic cancer tissue. This probe does not require conjugation for it to target pancreatic cancer tissue, and it allows for visualization at four levels of structure – whole organ, resected tissue, individual cells, and subcellular organelles. Their work appears in ACS Omega.
"The high selectivity of the fluorescent probe can help mitigate subjectivity involved in analyzing frozen sections," says co-author Professor Robert M. Strongin of Portland State University. "It is the only synthetic probe that we are aware of that enables analysis within a time that is short enough to be compatible with the operating room workflow."
In the current study, Wang et al. sought to develop a fluorescent probe whose excitation and emission maxima were in the near infrared range (NIR, 650 to 900 nm). Fluorophores in the NIR range are advantageous for several reasons: 1) this range is where hemoglobin and water have the lowest absorption coefficients, 2) NIR causes little-to-no damage to bio samples, 3) NIR has good tissue penetration depth, and 4) NIR has a good signal-to-noise ratio.
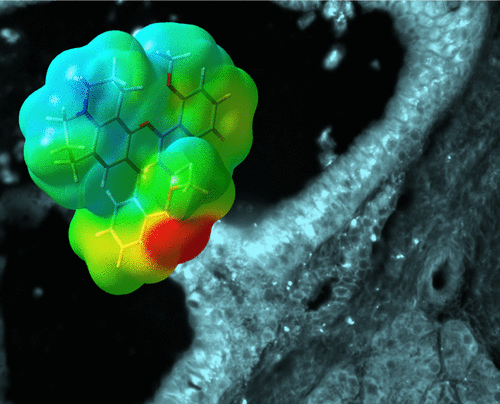
In searching for a fluorophore probe, Wang et al. began by making a library of systematically modified 1-substituted benzo[c]xanthenes. These structural scaffolds were chosen because they have several of the desired properties for targeted drugs (i.e., they satisfy the Lipinki Rule of Five and the Veber Rules). Additionally, the scaffolds fluoresce in the NIR range. They modified these scaffolds with hydroxy, methoxy, amine, and alkyl amine substituents. They tested each modified 1-substituted benzo[c]xanthene for how their emission spectra change based on solvent dependence and pH sensitivity.
From the thirteen compounds tested, three were selected as representative of the three classes of molecules they investigated (semi naphthofluorescein, semi naphthorhodamine, and semi naphthorhadafluor). In vitro tests were conducted using Capan-1 cells, which serve as a representative pancreatic ductal adenocarcinoma (PDAC) cell line. Of the three compounds tested, the semi naphthofluorescein scaffold with a methoxy substituent was the least toxic and showed the best localization within the cells.
The three compounds were then tested in vivo in healthy CD-1 mice. The first test was to see how the fluorescent probe distributed in healthy mice compared to methylene blue, an FDA-approved fluorescence agent for in vivo testing. Compared to methylene blue, both the semi naphthofluorescein with a methoxy substituent and the semi naphthorhodamine scaffold with a methoxy substituent exhibited better pancreatic tissue specificity.
The next step was to test the best fluorescent probe with genetically engineered mice with PDAC tumors that mimic human PDAC. The semi naphthorhodamine compound had the highest pancreatic tissue uptake in the healthy mice so it was tested in five genetically engineered mice.
The fluorescent probe worked well in the mice with PDAC. It targeted malignant cells compared to how the probe dispersed in healthy mice. Notably the fluorescent probe collected in ductal tissue epithelial cells. Pancreatic ductal epithelial cells are known to give rise to PDAC. Also the fluorescent probe accumulated in acinar cells. It has been shown that mutations in acinar can eventually lead to PDAC.
This study demonstrates a fluorescent probe that works well for high contrast studies for pancreatic cancer tissue identification both in vivo and ex vivo, and can serve as a faster, more accurate detection method for pancreatic cancer, particularly during surgical procedures.
"We plan to test human tissue and to evaluate the probe's utility in the operating room," Professor Strongin said. "Future studies also include the optimization of near infra-red fluorescent probe structure to further elucidate the physicochemical parameters that enable PDAC targeting to guide next generation probe and drug design."
More information: Lei Wang et al. Far-Red and Near-Infrared Seminaphthofluorophores for Targeted Pancreatic Cancer Imaging, ACS Omega (2017). DOI: 10.1021/acsomega.6b00403
Abstract
Molecular probes that selectively highlight pancreatic cancer (PC) tissue have the potential to improve pancreatic ductal adenocarcinoma (PDAC) margin assessment through the selective highlighting of individual PC cells. Herein, we report a simple and unique family of systematically modified red and near-infrared fluorescent probes that exhibit a field-effect-derived redshift. Two of thirteen probes distributed to the normal mouse pancreas following systemic administration. One selectively accumulated in genetically modified mouse models of PDAC. The probe exhibited intracellular accumulation and enabled visualization of four levels of the structure, including the whole organ, resected tissue, individual cells, and subcellular organelles. In contrast to the small-molecule probes reported previously, it possesses an inherent affinity toward PDAC cells and thus does not require conjugation to any targeting agent. The fluorescent probe can thus promote new strategies not only for precision image-guided surgery, but also for PC detection, monitoring of therapeutic outcomes, and basic research.
© 2017 Phys.org