Study finds key mechanism important for sex determination in vertebrates
In various vertebrate species, including fish, amphibians, reptiles, birds and mammals, the DMRT1 gene is a master sex regulator, working to develop and maintain 'maleness' -either the sex cells (or germline) into testis and/or maintaining masculinization throughout the other cells in the body (i.e., Latin for soma, or somatic cells). But little is known about the how the DMRT1 gene is turned on and off in vertebrate testicular somatic cells or germ cells.
Now, in a new study appearing in the advanced online edition of the journal Molecular Biology and Evolution, Michihiko Ito et al. have expanded the study of DMRT1 among the sea lamprey, the most primitive of vertebrates, the gecko reptile, and measured its regulation in a couple of frog species.
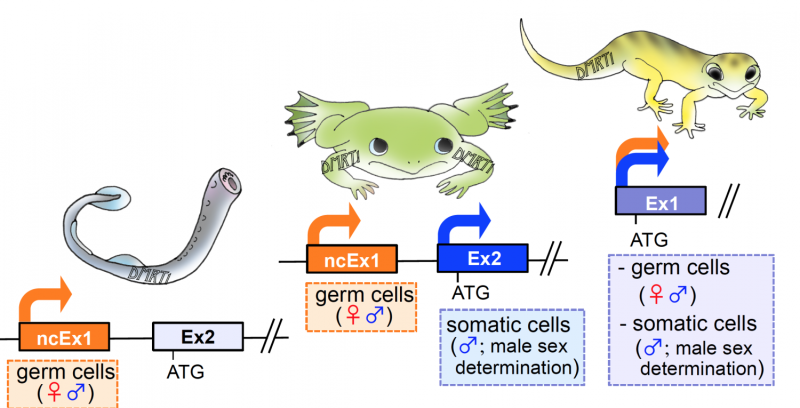
A comparison of the key regulator region of DMRT1 (called the ncEx1 promoter sequence, for their ability to promote, or turn on or off gene expression). They found the presence of ncEx1 in the lamprey dmrt1 gene, but not in the reptilian gecko.
"These results, taken together with our earlier findings, indicated that the ncEx1 of dmrt1 may have been lost in the common ancestor of reptiles and mammals," said corresponding author Michihiko Ito.
Next, two amphibian frog species, X. laevis and S. tropicalis, had their dmrt1 genes examined and were found to have two distinct promoters, one located upstream of the ncEx1 and the other located upstream of the translation start site-containing exon 2. They also found that the first and second promoters in X. laevis regulated the germ-cell- and somatic-cell-specific expression of dmrt1, respectively.
Furthermore, the analysis of X. laevis individuals with reduced expression of ncEx1-derived transcripts indicated an important role for DMRT1 in germ-cell development. The results indicated that the completely eliminating DMRT1 of the significantly decreased the number of germ cells in the gonads. In addition, three conserved noncoding sequences (CNSs) were identified among vertebrate DMRT1 genes.
The research team proposed a new model of the key mechanism behind transcriptional regulation of dmrt1 in germ and somatic cells by these promoters and three conserved noncoding sequences during vertebrate evolution.
"In a common ancestor of reptiles and mammals, one of the two promoters evolved into a dual promoter, capable of regulating both the somatic- and germ-cell expression of Dmrt1," said Ito. "We could not know how the one promoter got the dual functions. Therefore, the dual functions must be involved in germ cell- or somatic cell-related enhancer(s). Taken together, our findings suggest that dmrt1 regulates both gonadal somatic-cell masculinization and feminization by controlling germ-cell fate."
More information: Molecular Biology And Evolution (2016). DOI: 10.1093/molbev/msw273
Journal information: Molecular Biology and Evolution
Provided by Oxford University Press